First Efficacy Results from Africa find Typhoid Vaccine to offer 84 Percent Protection against Typhoid Fever
September 16, 2021 | January Payne

Only Typhoid Vaccine Proven Safe for Infants and Young Children
A new study, published in the New England Journal of Medicine, finds a single dose of typhoid conjugate vaccine (TCV) – the only typhoid vaccine licensed for children as young as 6 months – is safe and 84 percent effective in protecting against typhoid in Blantyre, Malawi. These are the first efficacy results from Africa and part of a five-year, multi-country project to accelerate introduction of TCV.
The findings are critical as typhoid is an increasing public health threat across sub-Saharan Africa due to the emergence and spread of multi-drug resistant strains, which are now common in Malawi. An estimated 1.2 million typhoid cases and 19,000 deaths occur each year in sub-Saharan Africa, which includes Malawi. The vast majority of these infections occur in school-age and pre-school children.
The study is being conducted through a partnership between the University of Maryland School of Medicine’s (UMSOM) Center for Vaccine Development and Global Health (CVD), the Malawi-Liverpool-Wellcome Trust (MLW) and Blantyre Malaria Project (BMP) in Blantyre, Malawi.
 The work is part of the Typhoid Vaccine Acceleration Consortium (TyVAC) – a larger effort led by CVD, the Oxford Vaccine Group at the University of Oxford, and PATH. TyVAC aims to accelerate the introduction of TCV as part of an integrated approach to reduce the burden of typhoid in countries eligible for support from Gavi, the Vaccine Alliance.
The work is part of the Typhoid Vaccine Acceleration Consortium (TyVAC) – a larger effort led by CVD, the Oxford Vaccine Group at the University of Oxford, and PATH. TyVAC aims to accelerate the introduction of TCV as part of an integrated approach to reduce the burden of typhoid in countries eligible for support from Gavi, the Vaccine Alliance.
 In 2017, the World Health Organization (WHO) recommended TCV for children six months of age and older in typhoid endemic settings.
In 2017, the World Health Organization (WHO) recommended TCV for children six months of age and older in typhoid endemic settings.
The new study found that the vaccine prevented typhoid in Malawi – where the incidence of typhoid is high. This helps to pave the way for additional countries to introduce TCV, making it available to all children.
“Our only real option to controlling these new resistant strains of typhoid in a timely way is through the vaccine,” said Professor Melita Gordon, University of Liverpool, MLW, and principal investigator for the Malawi study. “The TCV efficacy data are the first from Africa and offer great promise for the control of this deadly disease across the continent.”
The analysis included more than 28,000 children aged 9 months to 12 years in Blantyre, Malawi – half received TCV and half received the control, group-A meningococcal (MenA) vaccine. Blood tests performed on children with febrile illness occurring at least two weeks after receiving a study vaccine confirmed typhoid in 10 children in the TCV group and 61 who received MenA. TCV was effective in all age groups, including children under five years of age.
Typhoid is a bacterial infection caused by Salmonella enterica serovar Typhi and is a serious threat in many low- and middle-income countries. While typhoid is treatable, the effects can go beyond illness and death. Typhoid can impair physical and cognitive development in children, affect school attendance and performance, limit productivity, and reduce earning potential.
 “TCVs have the potential to protect millions of children disproportionately impacted by typhoid. We hope these much-needed data catalyze additional countries to introduce this lifesaving vaccine,” said Kathleen Neuzil, MD, MPH, the Myron M. Levine, MD, DTPH, Professor in Vaccinology at UMSOM. Dr. Neuzil is also the director of CVD and principal investigator for TyVAC.
“TCVs have the potential to protect millions of children disproportionately impacted by typhoid. We hope these much-needed data catalyze additional countries to introduce this lifesaving vaccine,” said Kathleen Neuzil, MD, MPH, the Myron M. Levine, MD, DTPH, Professor in Vaccinology at UMSOM. Dr. Neuzil is also the director of CVD and principal investigator for TyVAC.
Prevention of typhoid – with vaccines – is critical amid rising antimicrobial resistance. The TyVAC study in Africa helps inform a country’s decision to introduce TCV into their national childhood immunization program to combat the growing threat of antimicrobial resistance occurring globally.
“These findings highlight the burden of typhoid illness and the remarkable impact of a single dose of TCV in improving the health of children in low- and middle-income countries,” said E. Albert Reece, MD, PhD, MBA, Executive Vice President for Medical Affairs, UM Baltimore, and the John Z. and Akiko K. Bowers Distinguished Professor and Dean, UMSOM. “CVD has again harnessed the power of partnership to address the globe’s most pressing problems.”
TyVAC is funded by the Bill & Melinda Gates Foundation. Typbar TCV® is licensed by Bharat Biotech International Limited, Hyderabad, India.
About the Center for Vaccine Development and Global Health at the University of Maryland School of Medicine
For over 40 years, researchers in the Center for Vaccine Development and Global Health (CVD) have worked domestically and internationally to develop, test, and deploy vaccines to aid the world’s underserved populations. CVD is an academic enterprise engaged in the full range of infectious disease intervention from basic laboratory research through vaccine development, pre-clinical and clinical evaluation, large-scale pre-licensure field studies, and post-licensure assessments. CVD has created and tested vaccines against cholera, typhoid fever, paratyphoid fever, non-typhoidal Salmonella disease, shigellosis (bacillary dysentery), Escherichia coli diarrhea, nosocomial pathogens, tularemia, influenza, coronaviruses, malaria, and other infectious diseases. CVD’s research covers the broader goal of improving global health by conducting innovative, leading research in Baltimore and around the world. Our researchers are developing new and improved ways to diagnose, prevent, treat, control, and eliminate diseases of global impact, including COVID-19. In addition, CVD’s work focuses on the ever-growing challenge of antimicrobial resistance.
About the University of Maryland School of Medicine
Now in its third century, the University of Maryland School of Medicine was chartered in 1807 as the first public medical school in the United States. It continues today as one of the fastest growing, top-tier biomedical research enterprises in the world -- with 46 academic departments, centers, institutes, and programs, and a faculty of more than 3,000 physicians, scientists, and allied health professionals, including members of the National Academy of Medicine and the National Academy of Sciences, and a distinguished two-time winner of the Albert E. Lasker Award in Medical Research. With an operating budget of more than $1.2 billion, the School of Medicine works closely in partnership with the University of Maryland Medical Center and Medical System to provide research-intensive, academic and clinically based care for nearly 2 million patients each year. The School of Medicine has nearly $600 million in extramural funding, with most of its academic departments highly ranked among all medical schools in the nation in research funding. As one of the seven professional schools that make up the University of Maryland, Baltimore campus, the School of Medicine has a total population of nearly 9,000 faculty and staff, including 2,500 students, trainees, residents, and fellows. The combined School of Medicine and Medical System (“University of Maryland Medicine”) has an annual budget of over $6 billion and an economic impact of nearly $20 billion on the state and local community. The School of Medicine, which ranks as the 8th highest among public medical schools in research productivity (according to the Association of American Medical Colleges profile) is an innovator in translational medicine, with 606 active patents and 52 start-up companies. In the latest U.S. News & World Report ranking of the Best Medical Schools, published in 2021, the UM School of Medicine is ranked #9 among the 92 public medical schools in the U.S., and in the top 15 percent (#27) of all 192 public and private U.S. medical schools. The School of Medicine works locally, nationally, and globally, with research and treatment facilities in 36 countries around the world. Visit medschool.umaryland.edu
Contact
January Payne
Director of Public Relations
(443) 203-8183
january.payne@som.umaryland.edu
Related stories

Wednesday, October 08, 2025
New Vaccine Shows Promise Against Typhoid and Invasive Salmonella in First Human Trial
Researchers at the University of Maryland School of Medicine’s Center for Vaccine Development and Global Health (CVD) have completed a successful Phase 1 clinical trial of a novel vaccine designed to protect against both typhoid fever and invasive non-typhoidal Salmonella--two major causes of illness and death among children in sub-Saharan Africa.

Wednesday, March 12, 2025
Meningococcal Vaccine Found to be Safe and Effective for Infants in Sub-Saharan Africa
University of Maryland School of Medicine (UMSOM) researchers helped conduct an important new global health study that found a vaccine that protects against five strains of meningitis prevalent in sub-Saharan Africa is safe and effective for use in young children beginning at 9 months of age. This study provided evidence that formed the basis for the World Health Organization’s (WHO) decision last year to recommend the pentavalent Men5CV meningitis vaccine for infants ages 9 months and older.

Tuesday, April 30, 2024
Miriam K. Laufer Appointed Interim Director of the Center for Vaccine Development and Global Health
University of Maryland School of Medicine (UMSOM) Dean, Mark T. Gladwin, MD, announced that Miriam K. Laufer, MD, Professor of Pediatrics, Medicine, and Epidemiology & Public Health, has been appointed as the Interim Head of UMSOM's Center for Vaccine Development and Global Health (CVD).

Thursday, March 28, 2024
NIH selects Dr. Kathleen Neuzil as Director of The Fogarty International Center
Kathleen M. Neuzil, MD, MPH, Director of the University of Maryland School of Medicine’s (UMSOM) Center for Vaccine Development and Global Health, has been named the 13th director of the Fogarty International Center (FIC), which is part of the National Institutes of Health (NIH). Dr. Neuzil will be the first woman to hold the permanent directorship since the center’s founding in 1968 and will also hold the position of Associate Director for International Research at NIH.
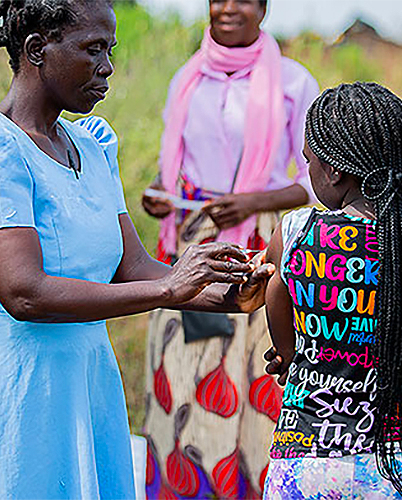
Thursday, January 25, 2024
Single Dose Typhoid Conjugate Vaccine (TCV) Provides Lasting Efficacy in Children
A single dose of the typhoid conjugate vaccine, Typbar TCV, provides lasting efficacy in preventing typhoid fever in children ages 9 months to 12 years old, according to a new study conducted by researchers at University of Maryland School of Medicine’s (UMSOM) Center for Vaccine Development and Global Health (CVD) and led by in-country partners at the Malawi-Liverpool Wellcome Trust (MLW) Clinical Research Programme.

Thursday, October 12, 2023
UM School of Medicine's Kirsten Lyke Elected as Member of Prestigious National Academy of Medicine
Kirsten E. Lyke, MD, Professor of Medicine and Physician-Scientist at the Center for Vaccine Development and Global Health (CVD) at the University of Maryland School of Medicine (UMSOM), was elected this week as a new member of the National Academy of Medicine (NAM). She was recognized for her pivotal research in emerging infections and human challenge models that have informed and shaped global vaccine and public health policy.

Monday, October 02, 2023
UM School of Medicine Researchers Present Interim Results on Meningococcal Vaccine for Infants and Young Children in Africa
University of Maryland School of Medicine (UMSOM) researchers, as part of the Infectious Diseases Clinical Research Consortium (IDCRC), provided an interim analysis showing that the pentavalent (NmCV-5) meningitis vaccine is safe for use in 9-month-old infants in the meningitis belt of sub-Saharan Africa. They presented their results to the World Health Organization’s (WHO) Strategic Advisory Group of Experts (SAGE) on Immunization on September 26.

Thursday, January 26, 2023
Small Study Shows Promise for Antimalarial Monoclonal Antibody to Prevent Malaria
A monoclonal antibody treatment was found to be safe, well tolerated, and effective in protecting against malaria in a small group of healthy volunteers who were exposed to malaria in a challenge study, according to new research published in by researchers at the University of Maryland School of Medicine (UMSOM).
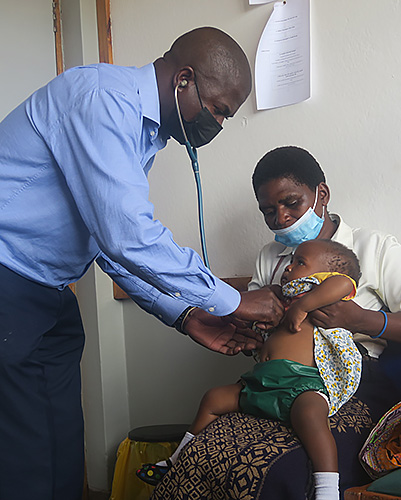
Tuesday, August 09, 2022
New Study Confirms Typhoid Vaccine Safety, Immune Response in Children
A new study, published in The Lancet Global Health, finds typhoid conjugate vaccine, Typbar TCV®, provides immunity for up to 3 years in children as young as 9 months old in Malawi. The research – conducted by the Blantyre Malaria Project, Malawi-Liverpool-Wellcome Trust, and researchers at the Center for Vaccine Development and Global Health (CVD) at the University of Maryland School of Medicine (UMSOM) – found that the TCV vaccine is safe and well tolerated. Importantly, the vaccine can be given to 9-month-old infants at the same time as routine measles-rubella vaccinations without reducing the immune response to either vaccine.

Monday, August 08, 2022
New Study Finds Rapid Decline in Vaccine-Boosted Neutralizing Antibodies Against Omicron Subvariant BA.5
A study led in part by investigators at the University of Maryland School of Medicine’s (UMSOM) Center for Vaccine Development and Global Health found that although COVID-19 booster vaccinations in adults elicit high levels of neutralizing antibodies against the Omicron variant of SARS-CoV-2, those antibody levels decrease substantially within three months. Kirsten E. Lyke, MD, Professor of Medicine at UMSOM and scientist at CVD, is Co-Chair and site Principal Investigator for the study, and Meagan Deming, MD, PhD, Assistant Professor of Medicine at the UMSOM, also a scientist at CVD, is Vice-Chair of the study, which is a collaboration between investigators at the UMSOM’s CVD and the Institute of Human Virology (IHV).
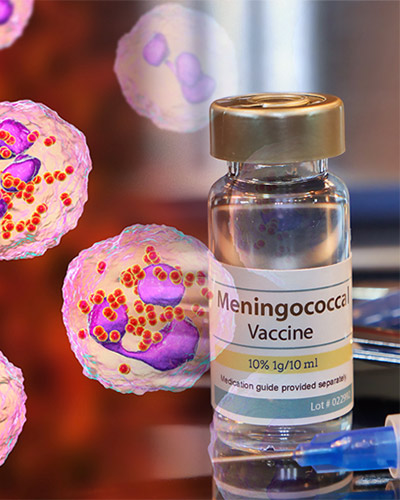
Thursday, March 24, 2022
UM School of Medicine Leads Research to Assess Meningococcal Vaccine for Infants and Young Children in Africa
Researchers at the University of Maryland School of Medicine (UMSOM)’s Center for Vaccine Development & Global Health (CVD) are leading a study to evaluate the use of a pentavalent – or five in one – meningococcal conjugate vaccine (NmCV-5) among infants and young children in the meningitis belt of sub-Saharan Africa. This is the final and pivotal study for World Health Organization (WHO) prequalification of this vaccine, which is the last stage to make the vaccine available for low- and middle-income countries.

Wednesday, January 27, 2021
Dr. Wilbur Chen, Nationally-Recognized Vaccine Researcher, Selected for Federal Committee that Guides Immunization Policies
Wilbur H. Chen, MD, MS, FIDSA, FACP, Professor of Medicine at the University of Maryland School of Medicine (UMSOM), has been named a new voting member of the federal government’s Advisory Committee on Immunization Practices (ACIP), the prestigious board of experts that makes recommendations on the safe use of vaccines for Americans. The U.S. Department of Health and Human Services selected Dr. Chen for the 15-member advisory committee based on his expertise and national leadership in vaccinology, infectious diseases, public health, and preventive medicine. He will remain in his current role at UMSOM while he serves in his four-year term, which began last month.
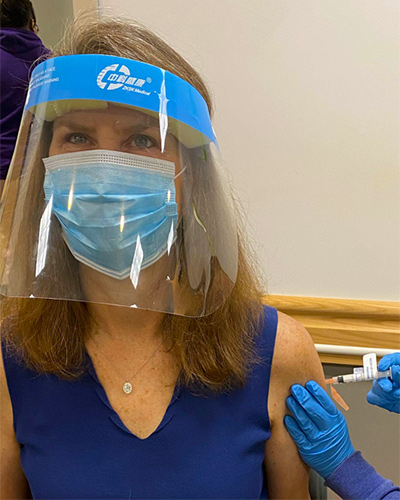
Tuesday, January 05, 2021
Dr. Kathleen Neuzil, World-renowned Leader in Vaccine Research, Receives Moderna Vaccine
Kathleen Neuzil, MD, MPH, FIDSA, the Myron M. Levine, MD, DTPH, Professor of Vaccinology and Director of the University of Maryland School of Medicine’s (UMSOM)’s Center for Vaccine Development and Global Health (CVD), received her first injection of the Moderna vaccine for COVID-19 on December 31. Researchers at the University of Maryland School of Medicine (UMSOM) played an integral part in the dedicated work that led to the U.S. Food and Drug Administration issuing an Emergency Use Authorization for the Moderna vaccine in December.

Tuesday, January 05, 2021
University of Maryland School of Medicine Begins Phase 3 Trial of Novavax COVID-19 Vaccine Candidate
Researchers at the University of Maryland School of Medicine (UMSOM) will participate in a Phase 3 clinical trial of an investigational COVID-19 vaccine to protect against SARS-CoV-2, the coronavirus causing COVID-19 that continues to impact millions of people around the world. The clinical trial will test the safety and effectiveness of NVX-CoV2373, being developed by U.S. biotechnology company, Novavax, Inc., based in Gaithersburg, MD.
Tuesday, May 05, 2020
UM School of Medicine is First in U.S. to Test Unique RNA Vaccine Candidate for COVID-19
In a significant development in the global effort to discover a safe and effective vaccine for COVID-19, researchers at the University of Maryland School of Medicine (UMSOM) became the first in the U.S. to begin testing experimental COVID-19 vaccine candidates developed by Pfizer and BioNTech. The research, funded by Pfizer Inc., will study the safety, efficacy, and dosing of an experimental mRNA -based vaccine.

Tuesday, March 20, 2018
UMSOM Vaccine Experts Lead Trial on Avian Flu Vaccine
Vaccine experts at the University of Maryland School of Medicine (UMSOM) have begun multiple clinical trials of vaccines designed to protect against H7N9, an avian influenza virus that was first reported in humans in 2013 in China.

Wednesday, August 02, 2017
UM School of Medicine Receives $2 Million Grant for HIV Research in Malawi
The University of Maryland School of Medicine’s (UM SOM) Institute for Global Health (IGH) and the Institute of Human Virology (IHV) have been awarded a $2 million five-year grant from the Eunice Kennedy Shriver National Institute of Child Health and Human Development to study the impact exposure to HIV has on the immune systems of infants in utero and how those changes impact the ability of infants to fight off infections after birth.

Monday, May 15, 2017
UM SOM Vaccine Researcher Receives Top Award in the Study of Infectious Diseases
Myron M. Levine, MD, DTPH, the Simon and Bessie Grollman Distinguished Professor and Associate Dean for Global Health, Vaccinology and Infectious Disease at UM SOM, has been awarded the Maxwell Finland Award for Scientific Achievement by the National Foundation for Infectious Diseases (NFID). The award honors his extensive accomplishments in public health; over his career, he has identified solutions to major sources of disease in the developing world, including cholera, typhoid, and Shigella dysentery. The award will be presented on Thursday, May 18, 2017 at the Hyatt Regency Bethesda in Bethesda, Md.

Friday, December 23, 2016
Trial Results Confirm Ebola Vaccine Provides High Protection Against Disease
An experimental Ebola vaccine was highly protective against the deadly virus in a major trial in Guinea, according to a new study that included researchers from the University of Maryland School of Medicine (UM SOM).

Monday, January 11, 2016
UM SOM to Collaborate with Industry to Develop a Vaccine to Prevent Common, Deadly Infection
The Center for Vaccine Development (CVD) at the University of Maryland School of Medicine (UM SOM) will participate in a partnership with industry to develop a vaccine to prevent a group of deadly bacterial infections that occur commonly among hospital patients
