August 04, 2016
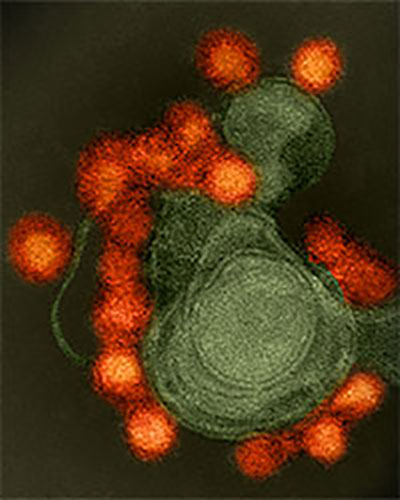
UM SOM'S Center for Vaccine Development, Tapped Previously for Ebola Vaccine, Now Steps Up to Help Test NIH-Developed Zika Vaccine
As world leaders increasingly recognize the Zika virus as an international public health threat, the Center for Vaccine Development (CVD) at the University of Maryland School of Medicine's Institute for Global Health (IGH) has been chosen as one of three study sites in a human safety trial of a new Zika vaccine. The early-stage study will evaluate the experimental vaccine's safety and ability to generate an immune system response in participants.
The selection of the University of Maryland School of Medicine (UM SOM) marks the second time in two years that the School's internationally-acclaimed CVD has been tapped to assist in vaccine testing efforts amidst a growing crisis. In 2014, the UM SOM was the only U.S. medical school asked to join an unprecedented international consortium formed by the World Health Organization. The Consortium resulted in the evaluation of one of the first experimental Ebola vaccines (ChAd3-EBO-Z), developed by the Vaccine Research Center (VRC) of the National Institute of Allergy and Infectious Diseases (NIAID). Recently, the CVD received FDA approval for the first vaccine approved in the U.S. for protection against Cholera. In July, the CVD began malaria vaccine trials in Burkina Faso.

The Zika vaccine trial, which will involve at least at least 80 volunteers at three sites in the United States, is being undertaken by NIAID, part of the National Institutes of Health (NIH). The early-stage study will evaluate the experimental vaccine's safety and ability to generate an immune system response against Zika.
Scientists at NIAID's VRC developed the investigational vaccine—called the NIAID Zika virus investigational DNA vaccine—earlier this year.
The Principal Investigator on the study is Monica A.  McArthur, MD, PhD, Assistant Professor, Department of Pediatrics at the UM SOM, whose primary research interests are the interactions between pathogens and the human immune system.
McArthur, MD, PhD, Assistant Professor, Department of Pediatrics at the UM SOM, whose primary research interests are the interactions between pathogens and the human immune system.
"As we learn more about the threat that Zika poses, and as it spreads further and further, the need for a vaccine becomes greater," said Kathleen Neuzil, MD, MPH, CVD Director, Professor of Medicine, and Deputy Director of IGH "The CVD welcomes the opportunity to collaborate with the NIH on this extremely important effort."
The study is part of the U.S. government response to the ongoing outbreak of Zika virus in the Americas. According to the Centers for Disease Control and Prevention, more than 50 countries and territories have active Zika virus transmission. In the United States and its territories, more than 6,400 Zika cases have been reported. Although Zika infections are usually asymptomatic, some people experience mild illness lasting about a week. However, Zika virus infection during pregnancy can cause a serious birth defect called microcephaly, as well as other severe fetal defects of the brain and other organs. There are no vaccines or specific therapeutics to prevent or treat Zika virus disease.
The NIAID Zika virus investigational DNA vaccine approach is similar to that used for another investigational vaccine developed by NIAID for West Nile virus. That vaccine candidate was found to be safe and induced an immune response when tested in a Phase 1 clinical trial.
The investigational Zika vaccine includes a small, circular piece of DNA—called a plasmid—that scientists engineered to contain genes that code for proteins of the Zika virus. When the vaccine is injected into the arm muscle, cells read the genes and make Zika virus proteins, which self-assemble into virus-like particles. The body mounts an immune response to these particles, including neutralizing antibodies and T cells. DNA vaccines do not contain infectious material—so they cannot cause a vaccinated individual to become infected with Zika—and have been shown to be safe in previous clinical trials for other diseases.
"A team of scientists here at NIAID worked tirelessly to rapidly develop this vaccine for clinical testing," said John Mascola, MD, director of NIAID's VRC. "DNA or gene-based vaccines induce antibodies, but they also can activate the cell-mediated immune response, which ultimately could yield strong and durable protection against disease."
The Phase 1 clinical trial, called VRC 319, is led by Julie E. Ledgerwood, D.O., chief of the VRC's clinical trials program. Volunteers will be divided randomly into four study groups of 20 people each. After enrollment, all participants will receive a vaccination at their first visit via a needle-free injector that pushes the vaccine fluid into the arm muscle. Half of the participants will receive one additional vaccination eight weeks or 12 weeks later. The remaining participants will receive two additional vaccinations: one group of 20 participants will receive a second vaccine at week four and a third at week eight; the other group of 20 participants will receive a second vaccine at week four and a third at week 20. All participants will receive the same dose at each vaccination.
Following each vaccination, participants will remain at the study site for observation for a minimum of 30 minutes so clinicians can monitor for any adverse reactions. Participants will receive a diary card to use at home to record their temperature and any symptoms for seven days following each vaccination.
All participants will return for follow-up visits within a 44-week time period after the first vaccination so investigators can monitor their health to determine if the vaccine is safe. The study team will review patient data daily and weekly to monitor safety. A Protocol Safety Review Team will also conduct formal interim safety reviews.
At follow-up visits, investigators will also take blood samples for laboratory testing to measure the immune response to the vaccine. Participants will be asked to return for two follow-up visits at 18 months and two years following the initial vaccination so investigators can obtain additional blood samples to assess the durability of the immune response.
The other two study sites are Emory University in Atlanta and NIH in Bethesda, Maryland. Initial safety and immunogenicity data from the Phase 1 trial are expected by the end of 2016. If results show a favorable safety profile and immune response, NIAID plans to initiate a Phase 2 trial in Zika-endemic countries in early 2017.
"Zika is an urgent international public health threat, including, now, in parts of our own country," said UM SOM Dean E. Albert Reece, MD, PhD, MBA, who is also vice president for medical affairs at the University of Maryland and the John Z. and Akiko K. Bowers Distinguished Professor. "It is gratifying to see the University of Maryland School of Medicine's Center for Vaccine Development continue to be involved on the front lines of addressing these global threats, as we have done with Ebola and other infectious diseases."
About the University of Maryland School of Medicine
The University of Maryland School of Medicine was chartered in 1807 and is the first public medical school in the United States and continues today as an innovative leader in accelerating innovation and discovery in medicine. The School of Medicine is the founding school of the University of Maryland and is an integral part of the 11-campus University System of Maryland. Located on the University of Maryland's Baltimore campus, the School of Medicine works closely with the University of Maryland Medical Center and Medical System to provide a research-intensive, academic and clinically based education. With 43 academic departments, centers and institutes and a faculty of more than 3,000 physicians and research scientists plus more than $400 million in extramural funding, the School is regarded as one of the leading biomedical research institutions in the U.S. with top-tier faculty and programs in brain science, cancer, surgery and transplantation, trauma and emergency medicine, vaccine development and human genomics, among other centers of excellence. The School is not only concerned with the health of the citizens of Maryland and the nation, but also has a global presence, with research and treatment facilities in more than 35 countries around the world. http://medschool.umaryland.edu/
About the UM SOM CVD
The CVD, which was founded by Dr. Myron "Mike" Levine in 1974, is one of the most recognized entities for developing, testing, and implementing vaccines in the world. Within the IGH, the CVD develops and tests vaccines to prevent infectious diseases that disproportionately affect people living in the least developed countries. For the past 40 years, the CVD has conducted a wide range of research relating to the development of vaccines for a variety of diseases, including cholera, typhoid fever, paratyphoid fever, non-typhoidal Salmonella disease, shigellosis, Escherichia coli diarrhea, malaria, and other infectious diseases, including influenza. The CVD also developed new delivery systems, as well as public health and vaccine policy around the world, including Africa, Asia, and Latin America. Most recently, researchers at the CVD received worldwide attention for leading CVD's direct involvement in the WHO's global consortium for accelerated testing of a new Ebola vaccine candidate in Mali and Baltimore.
Contact
Office of Public Affairs
655 West Baltimore Street
Bressler Research Building 14-002
Baltimore, Maryland 21201-1559
Contact Media Relations
(410) 706-5260
Related stories

Thursday, July 02, 2020
UMSOM Researchers Help Weigh Role of Human Challenge Studies for COVID-19 Vaccine Development
Members of the National Institutes of Health’s (NIH) Accelerating COVID-19 Therapeutic Interventions and Vaccines (ACTIV) Working Group, which includes Kathleen Neuzil, MD, MPH, DTPH, the Myron M. Levine, MD, DTPH Professor in Vaccinology and Director of the Center for Vaccine Development and Global Health (CVD) at the University of Maryland School of Medicine (UMSOM), assessed the practical considerations and prerequisites for using controlled human infection models (CHIMs) to support development of a SARS-CoV-2 Vaccine.

Friday, September 20, 2019
UM School of Medicine's Center for Vaccine Development and Global Health Receives NIH Contract of up to More than $200 Million for Influenza Research
Kathleen Neuzil, MD, MPH, Professor of Medicine and Pediatrics and Director of the Center for Vaccine Development and Global Health (CVD) at the University of Maryland School of Medicine (UMSOM) and Dean E. Albert Reece, MD, PhD, MBA, announced that CVD has been awarded a contract from the National Institute of Allergy and Infectious Diseases (NIAID), with total funding up to more than $200 million over seven years if all contract options are exercised.

Friday, March 29, 2019
12th Annual Dr. Aaron I. Grollman Visiting Professorship in Basic Sciences Lectureship Features Dr. Eva Harris
Eva Harris, PhD, never met a mosquito she didn’t like. A Professor of Infectious Diseases and Vaccinology and Director of the Center for Global Health at the University of California, Berkley, Dr. Harris was recently the guest speaker at the University of Maryland School of Medicine’s (UMSOM) Dr. Aaron I. Grollman Visiting Professorship in Basic Sciences Lectureship, where she shared her research on the control of Zika, dengue, and chikungunya—the most prevalent mosquito-borne diseases in humans. She is also the Co-Founder and President of the Sustainable Sciences Institute (SSI), an international non-profit dedicated to developing sustainable local research and public health systems in resource-poor settings.
Friday, March 08, 2019
UMSOM Researchers Unveil Progress and Challenges in Introducing Typhoid Conjugate Vaccine in Sub-Saharan Africa and Asia
Each year there are nearly 11 million cases of typhoid, a disease that is spread through contaminated food, drink and water. Researchers at the University of Maryland School of Medicine are leading an international consortium that is studying the impact of a typhoid conjugate vaccine (TCV) in an effort to accelerate introduction of the vaccine in countries in sub-Saharan Africa and Asia where there is a high burden of typhoid.
Monday, December 03, 2018
UMSOM and Groupe De Recherche Action En Sante Begin Second Typhoid Conjugate Vaccine Study in Africa
A new study has been launched in Burkina Faso for Bharat Biotech’s typhoid conjugate vaccine (TCV). It is the second clinical study underway in Africa for the vaccine and the first in West Africa. The vaccine study is a joint effort by the Center for Vaccine Development and Global Health (CVD) at the University of Maryland School of Medicine, (UMSOM) and Groupe de Recherche Action en Santé (GRAS) in Burkina Faso.

Friday, November 02, 2018
UMSOM Global Health Expert Named to Prestigious World Health Organization Immunization Panel
Kathleen Neuzil, MD, MPH, Professor of Medicine and Pediatrics and Director of the Center for Vaccine Development and Global Health (CVD) at the University of Maryland School of Medicine (UMSOM), has been named to the World Health Organization’s (WHO) Strategic Advisory Group of Experts (SAGE) on Immunization.

Monday, April 23, 2018
UM School of Medicine Launches Newly Organized Center for Vaccine Development and Global Health Under Leadership of International Vaccine Expert Dr. Kathleen Neuzil
University of Maryland School of Medicine (UMSOM) Dean E. Albert Reece, MD, PhD, MBA, announced today the launch of a newly organized Center for Vaccine Development and Global Health (CVD). The new Center will be led by UMSOM Professor of Medicine and Pediatrics, Kathleen Neuzil, MD MPH, FIDSA, one of the world’s most influential research scientists and advocates in vaccine development and policy.

Thursday, February 01, 2018
UMSOM’s Monica McArthur, MD, PhD. Discusses Zika Virus and Challenges in Developing a Vaccine
Zika vaccine development: two years on from the outbreak

Thursday, September 28, 2017
UM SOM Vaccine Expert Warns of Risks of Influenza to Older Populations
“Protect yourself and others by getting the flu shot” was the message from Kathleen Neuzil, MD, MPH, FIDSA, Director of the Center for Vaccine Development (CVD) at the University of Maryland School of Medicine (UM SOM). Dr. Neuzil was among the speakers at a press conference hosted by the National Foundation for Infectious Diseases. U.S. Secretary of Health and Human Services, Thomas E Price, MD, also spoke at the press conference and urged people to get their flu shot. The panel members all received their flu shots following the press conference.
Monday, May 15, 2017
U.S. CDC Recommends Use of Cholera Vaccine Developed by University of Maryland School of Medicine
A cholera vaccine developed by scientists at the University of Maryland School of Medicine’s Center for Vaccine Development (CVD) has been recommended by the U.S. Centers for Disease Control and Prevention (CDC) for use as a protection for U.S. adults traveling to areas with cholera. The CDC’s latest recommendation was published on May 11, in Morbidity and Mortality Weekly Report.

Thursday, January 05, 2017
For the First Time, Researchers Identify Key Proteins That May Make Zika So Deadly
Until it burst onto the scene earlier this year, Zika was an obscure, little-known virus. As a result, scientists know little about how it works. Over the past year, they have learned that it can cause a range of dangerous health problems, including birth defects such as microcephaly and neurological problems such as Guillain-Barré syndrome. But they don’t know which Zika protein or proteins are causing harm, or exactly how these proteins cause damage.

Thursday, November 10, 2016
International Consortium Receives $36.9 Million Grant to Fight Typhoid
Typhoid fever, a bacterial infection that causes high fever and other disabling symptoms, remains a serious global problem in the developing world: it kills almost a quarter of a million people annually, and infects about 21 million.

Thursday, September 22, 2016
University of Maryland School of Medicine Holds Inaugural Global Health Summit
The Institute for Global Health (IGH) at the University of Maryland School of Medicine (UM SOM) will hold its first Global Health Summit on September 26. The event, which will take place from noon to 6 p.m. in the SMC Campus Center at 621 W. Lombard Street, will seek to foster collaboration among scientists and promote new and innovative global health research at UM SOM, as well as at other schools at the University of Maryland, Baltimore (UMB). Faculty and staff from UM SOM, as well as other schools are welcome at the event.

Wednesday, June 29, 2016
UM SOM Researchers Awarded Grant to Use Innovative Alternative to Autopsies to Better Understand Child Mortality
Kathleen Neuzil, MD, MPH, director of the Center for Vaccine Development (CVD) at the University of Maryland School of Medicine (UM SOM), and UM SOM Dean E. Albert Reece, MD, PhD, MBA, announced today that CVD has been awarded a large grant from the Bill & Melinda Gates Foundation for research that will help determine why so many children under five are dying in the world’s poorest countries. The grant will fund use of an innovative alternative to traditional autopsy known as minimally invasive tissue sampling. The technique, which involves the collection of tissue samples with fine needles, allows researchers to quickly identify the cause of death, and help illuminate ways to save lives and improve the health of children in these vulnerable areas.

Tuesday, April 12, 2016
UM SOM Researcher Dr. Kathleen Neuzil Honored by Vanderbilt University School of Medicine
The Vanderbilt University School of Medicine (VUSM) has announced that Kathleen Neuzil, MD, MPH, director of the Center for Vaccine Development at the University of Maryland School of Medicine (UM SOM), has received the 2016 Vanderbilt University School of Medicine Distinguished Alumni Award.

Tuesday, February 16, 2016
The Baltimore Sun: Baltimore Consortium Forms Global Task Force to Study Zika, Find Vaccine
A Baltimore-based research consortium is forming a task force of leading scientists from around the world, including renowned AIDS researcher Dr. Robert Gallo, to better understand the Zika virus and quickly develop a vaccine.
