New Study Provides Insight for How Congenital Heart Defects Manifest
July 29, 2022 | Vanessa McMains
University of Maryland School of Medicine findings demonstrate that cells lining the heart direct heart size
About one percent of the world population is born with a congenital heart defect, which affects about 40,000 U.S. births each year, but how these particular birth defects come about is largely unknown.
 In an effort to learn more about how the heart develops, researchers at the University of Maryland School of Medicine (UMSOM) have determined that the cells lining the heart direct the cardiac muscle to grow until the heart reaches its full size. They also outlined the complex mechanism that regulates this process, which requires two sets of brakes to be bypassed for the heart to develop properly.
In an effort to learn more about how the heart develops, researchers at the University of Maryland School of Medicine (UMSOM) have determined that the cells lining the heart direct the cardiac muscle to grow until the heart reaches its full size. They also outlined the complex mechanism that regulates this process, which requires two sets of brakes to be bypassed for the heart to develop properly.
The researchers say these findings explain a bit more about what can go wrong during development that can lead to heart birth defects and may also help develop better techniques for regenerating heart tissue.
“To recover from disease, you need to figure out how to do heart regeneration. At this time, no one can regenerate an entire heart, mostly because they have been focused on using the heart muscle to grow more heart muscle cells,” said Deqiang Li, PhD, Assistant Professor of Surgery at the University of Maryland School of Medicine in the Center for Vascular & Inflammatory Diseases. “Our findings suggest that we may need other cells from the heart, such as the epicardium (the cells lining the heart), to give the necessary instructions for the heart muscle to grow bigger.”
The mechanism the team described was published on June 20 in Circulation Research.
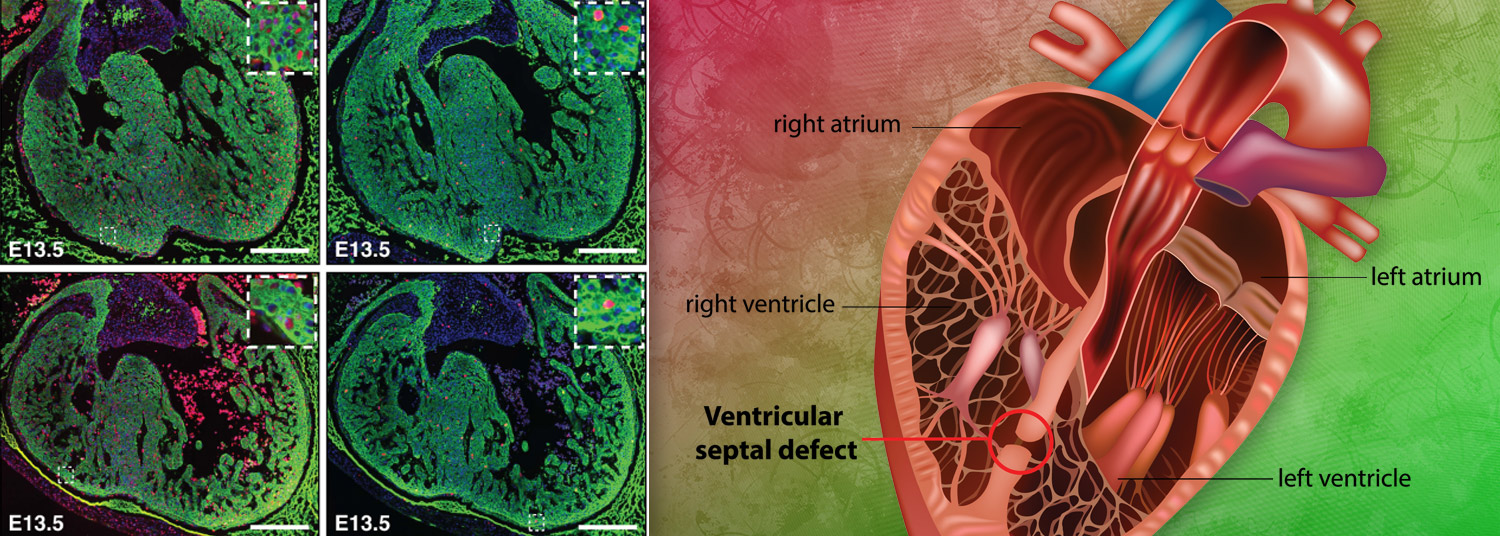
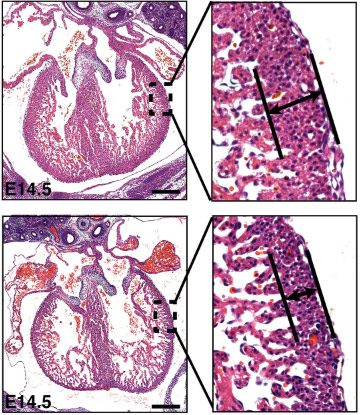
 The gene regulator histone deacetylase 3 (HDAC3, for short) was known to be important for development in the heart muscle cells, but whether it played specific role in the separate layer of cells lining the heart was unknown. To explore the role of HDAC3 in heart development, the researchers genetically engineered mice to lack HDAC3 only in the cells lining the heart. In fetal mice, they found that these hearts without HDAC3 in the heart lining had thinner, compact walls in the heart’s ventricles—basically, it seemed like the hearts did not grow enough.
The gene regulator histone deacetylase 3 (HDAC3, for short) was known to be important for development in the heart muscle cells, but whether it played specific role in the separate layer of cells lining the heart was unknown. To explore the role of HDAC3 in heart development, the researchers genetically engineered mice to lack HDAC3 only in the cells lining the heart. In fetal mice, they found that these hearts without HDAC3 in the heart lining had thinner, compact walls in the heart’s ventricles—basically, it seemed like the hearts did not grow enough.
The research team determined that the cells lining the heart without the gene regulator HDAC3 also made less of two growth factors that these cells normally pump out to encourage heart growth, while also making too much of two microRNAs. MicroRNAs are small pieces of genetic material that control what genes are turned on and made into proteins.
 “We were struggling in putting the pieces together for this mechanism for a long time. One day Postdoctoral Fellow and lead study author Jihyun Jang, PhD, approached me and expressed the brilliant idea of double brake mechanisms of the microRNAs preventing the growth factors from being made, which eventually turned out to be true!” said Dr. Li. “We would not have been able to complete this study without valuable contributions and insights from our co-authors, as well as support from the Department of Surgery and the Center for Vascular and Inflammatory Diseases.”
“We were struggling in putting the pieces together for this mechanism for a long time. One day Postdoctoral Fellow and lead study author Jihyun Jang, PhD, approached me and expressed the brilliant idea of double brake mechanisms of the microRNAs preventing the growth factors from being made, which eventually turned out to be true!” said Dr. Li. “We would not have been able to complete this study without valuable contributions and insights from our co-authors, as well as support from the Department of Surgery and the Center for Vascular and Inflammatory Diseases.”
Separately, they found that HDAC3 turns off genes that code for the two microRNAs, which allows the growth factors to be generated and ensures the heart grows to full-size.
“You may ask, why use such a complicated strategy that requires traversing two double brakes to get a normal heart to develop? Well, gene regulators like HDAC3 are found in every cell in the body, and microRNAs are also found everywhere. These specific regulatory hurdles allow this process to be specialized to various places in the body. Of course, that also means that these cell mechanisms may have applications for other diseases, as well, like cancer,” said Dr. Li. “To some people, this mechanism and these findings may seem incredibly detailed. If you think about life though, details matter. If one little thing messes up, then everything messes up.”
 E. Albert Reece, MD, PhD, MBA, Vice President for Medical Affairs, University of Maryland, and the John Z. and Akiko K. Bowers Distinguished Professor and Dean, University of Maryland School of Medicine, said, “One of the health conditions that I have spent much of my career studying is the mechanisms behind structural birth defects. Basic research, such as performed in this study, is essential for us to find out how the body develops normally, so that we can determine what goes wrong in disease, and ultimately one day, in this case, we can find ways to prevent congenital heart defects in the next generation of newborns.”
E. Albert Reece, MD, PhD, MBA, Vice President for Medical Affairs, University of Maryland, and the John Z. and Akiko K. Bowers Distinguished Professor and Dean, University of Maryland School of Medicine, said, “One of the health conditions that I have spent much of my career studying is the mechanisms behind structural birth defects. Basic research, such as performed in this study, is essential for us to find out how the body develops normally, so that we can determine what goes wrong in disease, and ultimately one day, in this case, we can find ways to prevent congenital heart defects in the next generation of newborns.”
Additional authors included Visiting Postdoctoral Fellow Guang Song, MD, PhD; Lab Technician Sarah Pettit; Postdoctoral Fellow Qinshan Li, MD, PhD; Visiting Student Xiaosu Song, MD, PhD; and Sunjay Kaushal, PhD, MD, Professor of Surgery, all of the University of Maryland School of Medicine; and Chen-leng Cai, PhD, of Indiana University.
This work was supported by the National Heart, Lung, and Blood Institute (grant R01HL153406) and startup funds from the Department of Surgery, University of Maryland School of Medicine.
About the University of Maryland School of Medicine
Now in its third century, the University of Maryland School of Medicine was chartered in 1807 as the first public medical school in the United States. It continues today as one of the fastest growing, top-tier biomedical research enterprises in the world — with 46 academic departments, centers, institutes, and programs, and a faculty of more than 3,000 physicians, scientists, and allied health professionals, including members of the National Academy of Medicine and the National Academy of Sciences, and a distinguished two-time winner of the Albert E. Lasker Award in Medical Research. With an operating budget of more than $1.3 billion, the School of Medicine works closely in partnership with the University of Maryland Medical Center and Medical System to provide research-intensive, academic, and clinically based care for nearly 2 million patients each year. The School of Medicine has nearly $600 million in extramural funding, with most of its academic departments highly ranked among all medical schools in the nation in research funding. As one of the seven professional schools that make up the University of Maryland, Baltimore campus, the School of Medicine has a total population of nearly 9,000 faculty and staff, including 2,500 students, trainees, residents, and fellows. The combined School of Medicine and Medical System (“University of Maryland Medicine”) has an annual budget of over $6 billion and an economic impact of nearly $20 billion on the state and local community. The School of Medicine, which ranks as the 8th highest among public medical schools in research productivity (according to the Association of American Medical Colleges profile) is an innovator in translational medicine, with 606 active patents and 52 start-up companies. In the latest U.S. News & World Report ranking of the Best Medical Schools, published in 2021, the UM School of Medicine is ranked #9 among the 92 public medical schools in the U.S., and in the top 15 percent (#27) of all 192 public and private U.S. medical schools. The School of Medicine works locally, nationally, and globally, with research and treatment facilities in 36 countries around the world. Visit medschool.umaryland.edu
Contact
Vanessa McMains
Director, Media & Public Affairs
University of Maryland School of Medicine
Institute of Human Virology
vmcmains@ihv.umaryland.edu
Cell: 443-875-6099
Related stories

Wednesday, February 22, 2023
The Molecular Mechanism Behind Abdominal Aneurysms
When the artery that supplies the stomach and the liver forms a bulge that ruptures, this medical emergency results in the deaths of 50 percent of patients before they reach the hospital. This “silent killer” condition, known as abdominal aortic aneurysm , led to the death of Albert Einstein — and is responsible for nearly 5,000 deaths in the U.S. each year. Now researchers have found new clues that eventually could lead to earlier detection methods to save lives in the future.