UM SOM Researchers Identify Potentially Revolutionary Antidepressant Compound
May 04, 2016
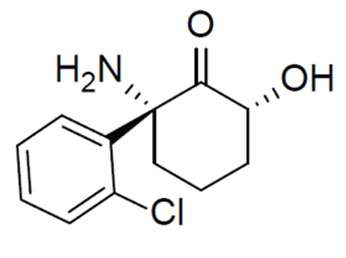
In Mice, Ketamine Metabolite Lifts Depression Quickly, Without Serious Side Effects
For years, scientists and doctors have known that ketamine can treat depression very rapidly, often working within hours, compared to weeks or months for widely used antidepressants. However, the drug, which is approved as an anesthetic, has major side effects – it is linked to hallucinations and dissociation - a sense of being outside your own body – and for these reasons is abused as a club drug. Not surprisingly, this limits its use in the treatment of depression.
A solution may be at hand. Researchers at the University of Maryland School of Medicine (UM SOM) have identified a metabolite of ketamine that quickly reverses depression in mice, but without ketamine's side effects. The study was published today in the journal Nature.
“This is potentially a major breakthrough,” said the study's senior author, Todd Gould, MD, an Associate Professor of Psychiatry at UM SOM. “It could allow depressed patients to get the rapid benefits of ketamine, while at the same time avoiding the risks.”
Most people with depression take medications that increase levels of the neurochemicals serotonin or norepinephrine in the brain. The most common of these drugs, such as Prozac and Lexapro, are selective serotonin reuptake inhibitors, or SSRIs. However, SSRIs are effective in only half of patients with depression, and even when these drugs work, they typically take between three and eight weeks to relieve symptoms. Ketamine, which does not work via serotonin or norepinephrine, can lift depression much more quickly, within hours after administration.
The work was a collaboration between scientists at UM SOM, the NIH National Institute of Mental Health (NIMH), the NIH National Center for Advancing Translational Sciences, the NIH National Institute on Aging, and the University of North Carolina.
“For years, we have been searching for ways to treat depression faster and more effectively,” said another co-author, Scott Thompson, PhD, Professor and Chair of the Department of Physiology at UM SOM, who has spent more than a decade studying glutamate and depression. “These results open up exciting new vistas for the first new generation of antidepressant compounds in the last 30 years.”
Using mice, the researchers tested the effects of several ketamine metabolites, chemicals produced by the breakdown of ketamine. They eventually focused on one called hydroxynorketamine, a compound that had been thought to have no psychoactive effects. In mice, blocking the transformation of ketamine to hydroxynorketamine prevented ketamine's antidepressant actions. Hydroxynorketamine itself also showed antidepressant effects, and none of ketamine's side effects.
“This discovery fundamentally changes our understanding of how this rapid antidepressant mechanism works, and holds promise for development of more robust and safer treatments," said Carlos Zarate, MD, of NIMH, a study co-author and a pioneer of research using ketamine to treat depression. "By using a team approach, researchers were able to reverse-engineer ketamine from the clinic to the lab to pinpoint what makes it so unique.”
The anesthetic and dissociative actions of ketamine are due to the fact that it blocks a particular glutamate receptor, the NMDA glutamate receptor. Researchers had assumed that ketamine acts as an antidepressant via the same mechanism. This appears not to be true, however, because hydroxynorketamine does not inhibit the NMDA receptor. Instead, hydroxynorketamine seems to activate another type of glutamate receptor, the AMPA receptor. Gould says that hydroxynorketamine likely works on depression via these AMPA receptors, either directly or indirectly.
Gould and his colleagues at NIH are already planning to begin testing hydroxynorketamine for its safety in humans. But he points out that the compound has, in effect, already been in humans in years as a metabolite following ketamine administration. "This gives us confidence it will be safe," said Gould.
“This study underscores the fact that our school is doing a range of important brain research. The Departments of Psychiatry, Physiology, and Neurology, as well as the Brain Science Research Consortium Unit and others, all are doing their part to advance our understanding of the brain and its diseases,” said UM SOM Dean E. Albert Reece, MD, PhD, MBA, who is also the vice president for Medical Affairs, University of Maryland, and the John Z. and Akiko K. Bowers Distinguished Professor. “In year to come, this finding may well change how depression is treated in millions of patients.”
About the University of Maryland School of Medicine
The University of Maryland School of Medicine was chartered in 1807 and is the first public medical school in the United States and continues today as an innovative leader in accelerating innovation and discovery in medicine. The School of Medicine is the founding school of the University of Maryland and is an integral part of the 11-campus University System of Maryland. Located on the University of Maryland’s Baltimore campus, the School of Medicine works closely with the University of Maryland Medical Center and Medical System to provide a research-intensive, academic and clinically based education. With 43 academic departments, centers and institutes and a faculty of more than 3,000 physicians and research scientists plus more than $400 million in extramural funding, the School is regarded as one of the leading biomedical research institutions in the U.S. with top-tier faculty and programs in cancer, brain science, surgery and transplantation, trauma and emergency medicine, vaccine development and human genomics, among other centers of excellence. The School is not only concerned with the health of the citizens of Maryland and the nation, but also has a global presence, with research and treatment facilities in more than 35 countries around the world. http://medschool.umaryland.edu/
Contact
Office of Public Affairs
655 West Baltimore Street
Bressler Research Building 14-002
Baltimore, Maryland 21201-1559
Contact Media Relations
(410) 706-5260
Related stories

Tuesday, August 30, 2022
Effects of Drugs in Mice can Depend on the Sex of the Human Experimenter
University of Maryland School of Medicine researchers demonstrated that a stress response in the brain is essential for ketamine’s antidepressant response in mice suggesting new ways to improve antidepressant therapy for patients who do not respond to the treatment.
Tuesday, April 13, 2021
UM School of Medicine Study Shows that Psychedelic Experience May Not be Required for Psilocybin’s Antidepressant-like Benefits
University of Maryland School of Medicine (UMSOM) researchers have shown that psilocybin—the active chemical in “magic mushrooms”— still works its antidepressant-like actions, at least in mice, even when the psychedelic experience is blocked. The new findings suggest that psychedelic drugs work in multiple ways in the brain, and it may be possible to deliver the fast-acting antidepressant therapeutic benefit without requiring daylong guided-therapy sessions.

Wednesday, March 27, 2019
UMSOM Researchers Discover a Critical Receptor Involved in the Response to Fast-Acting Antidepressants Like Ketamine
Effective treatment of clinical depression remains a major mental health issue, with roughly 30 percent of patients who do not respond to any of the available treatments. Researchers at the University of Maryland School of Medicine (UMSOM) have discovered a crucial receptor called mGlu2 that is critical to the mechanism of fast-acting antidepressants such as ketamine when used to treat depression.
