
The Typhoid Vaccine Acceleration Consortium (TyVAC) aims to accelerate the use of typhoid conjugate vaccine (TCV) in endemic settings and support country decision-making. We employ a complementary suite of activities to optimize the use – and ultimately public health impact – of TCVs while simultaneously supporting the long-term sustainability of TCV programs and collaborative partnerships.
Why Typhoid?
Typhoid, a serious enteric fever caused by Salmonella Typhi, is spread through contaminated food and water. While largely eliminated in industrialized countries, typhoid continues to be a substantial public health issue in Asia, sub-Saharan Africa, and parts of Latin America and the Middle East.
The Global Burden of Disease estimates typhoid caused more than 9 million infections and more than 110,000 deaths globally in 2019, with young children and adolescents aged 2 to 15 years disproportionately impacted. Though treatable with antibiotics, the number of cases resistant to available antibiotics is increasing.
There are currently four TCVs prequalified by the World Health Organization. Two – Typbar TCV® and TYPHIBEV® – are eligible for Gavi, the Vaccine Alliance support. A single dose of TCV is safe, well-tolerated, and efficacious for at least four years, including in children younger than 2 years of age.
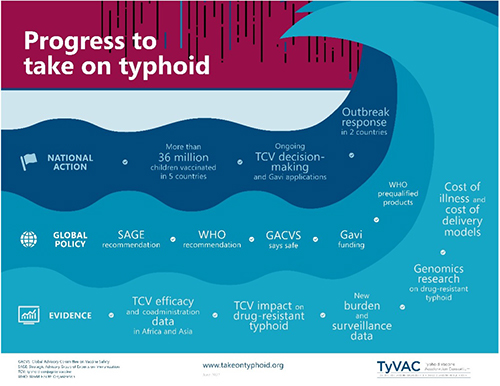
TyVAC's Integrated Approach
Engage with countries and local partners, provide assistance while fostering country ownership, strengthen local capacity, support decision-making, and enhance collaboration.
Coordinate with the typhoid community, maintain momentum and attention on typhoid as a global health priority, and ensure widespread and sustainable TCV introductions.
Generate new evidence related to typhoid disease burden, antimicrobial resistance, cost-effectiveness/cost-of-illness, and regional data on TCVs.
Advocate with partners for an integrated approach that includes vaccines in combination with improved water, sanitation, and hygiene (WASH).
Leadership

Dr. Matthew Laurens, Project Director, is a Professor of Pediatrics, Medicine, and Pathology at the University of Maryland School of Medicine. He has been an integral part of TyVAC since its inception, including working collaboratively with the teams in Malawi and Burkina Faso on clinical trials of TCV. Dr. Laurens is a member of the World Health Organization Technical Advisory Group on Salmonella Vaccines and served on the Typhoid Conjugate Vaccine Microarray Patch Full Vaccine Value Assessment Expert Group for the Vaccine Innovation Prioritization Strategy.
Former Director
Dr. Kathleen Neuzil was the Director of the Center for Vaccine Development and Global Health at the University of Maryland School of Medicine and the Myron M. Levine Professor in Vaccinology from 2015 through April 2024, when she assumed the role of Director, Fogarty International Center, NIH. She is an internationally recognized research scientist and advocate in the field of vaccinology and led TyVAC from its inception in 2017 through April 2024. Dr. Neuzil is a member of the World Health Organization Strategic Advisory Group of Experts on Immunization and the prestigious National Academy of Medicine.
Featured Blogs
Featured Blogs
- A mother’s battle [English Bangla]
- WASH and vaccines work together to fight disease
- Three things to know about typhoid and drug resistance
- Humanly possible: The people making vaccine introductions a reality [English Français]
Partners
 |
|---|
 |
|---|
Contact Us
- Phone: 410-706-5328
- Email: TyVAC@som.umaryland.edu
Follow Us
Visit the Take on Typhoid website for additional information and to sign up for our newsletter!


















