Search
Settings: systemic review and meta-analysis
Participants: 2 RCTs, 21 observational studies. Fifteen studies were published between 2020-2023.
There was a total of 25721 patients with septic shock
Outcome measurement: Primary outcome was short-term mortality (ICU, hospital, 28-day, 30-day). Secondary outcomes included ICU LOS, Hospital LOS, time to achieve MAP > 65 mm Hg,
Study Results:
Composite outcome of short term mortality:
- 20 studies and 17470 patients. Early initiation of vasopressors was associated with lower odds of short term mortality (OR 0.775, 95% CI 0.673-0.893, P<0.001, I2 = 68%).
- Early initiation of norepinephrine was associated with lower odds of short term mortality (OR 0.656, 95% CI = 0.544 to 0.790, P <0.001, I2 = 57.2%)
- Early initiation of vasopressin was also associated with lower odds of short term mortality (OR 0.685, 95% CI 0.558-0.840, P < 0.001, I2= 57%)
Secondary outcome:
- Early vasopressor group was associated with lower odds of RRT use (OR 0.796, 95% CI 0.654-0.968, P = 0.022, I2 = 0%)
- Mean Serum lactate levels at 6 hours was similar in early vasopressor group (Mean Difference 0.218, 95% CI -0.642 to 1.079, P = 0.619).
- However, mean serum lactate levels at 6 hours was lower in early norepinephrine subgroup (mean difference -0.489, 95% CI -0.863 to -0.115, P = 0.01).
Discussion:
- This appears to be a hot topic. When our group did this topic in 2020, there were 8 or 9 studies. Since 2020, there has been a significant increase in the number of publications, although most publications were observation studies.
- Early initiation of norepinephrine may reduce fluid overload, not by reducing fluid input, but by improving host inflammatory response, improving endothelial cell barrier stability.
- Counter-intuitively, early vasopressor was also found to be associated with lower incidence of arrhythmia, which the authors attributed to shorter duration of vasopressors and lower total dosage.
Conclusion:
More and more studies, although a RCT is still necessary, are showing that early initiation of vasopressor within 1-6 hours of septic shock would be more beneficial to patients with septic shock.
Show References
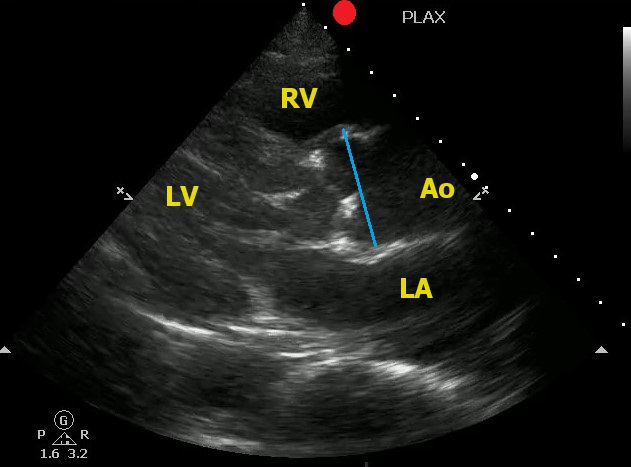
Point-of-Care Ultrasound can help to identify signs of thoracic aortic dissection.
One view to help in your assessment is the Parasternal Long Axis View.
- The aortic root should be in a 1:1:1 ratio with the left atrium and the right ventricle.
- The aortic root should be less than 4 cm (4.5 cm considered aneurysmal)
To correctly measure the aortic root:
- Measure at the Sinus of Valsalva
- Measure during diastole (when the aortic valve is closed)
- Measure leading edge to leading edge
Here is an example of an aortic root aneurysm:
Show References
A retrospective study of 2 years of data from 24 trauma centers looking at end tidal CO2 as a predictor of mortality in trauma patients found:
"A total of 1,324 patients were enrolled. ETCO2 was better in predicting mortality than shock index (SI) and systolic blood pressure (SBP). Prehospital lowest ETCO2 , SBP , and SI were all predictive of Mass Transfusion."
Another data point to consider when setting up trauma triage protocols and looking for patients who will require intensive interventions early.
Show References
The Bottom Line: Investigators studying the use of a pre-intubation checklist versus "usual care" found no differences in important outcomes such as oxygen saturation and first pass success. However, the study was conducted as a part of a larger study, was unblinded, and not well controlled.
The investigators who conducted the multicenter CHECK UP trial, a study of head up intubation in ICU patients, reviewed the care of 262 ICU patients who were intubated. Some intubation attempts were guided by a pre-intubation checklist and some were not. The authors found no difference between the groups in lowest SPO2, number of intubation attempts, etc. However, the study was unblinded and largely observational. In many cases, the elements of a checklist had been incorporated into routine practice.
Take Home Point: While the authors found no differences in outcomes, this study does little to prove or disprove the value of pre-intubation checklists. Not only was the study essentially uncontrolled, the untoward events being studied are unusual in the hands of experienced clinicians.
Comment: Pre-procedural checklists make intuitive sense to me. They help us to avoid cultural drift. I am certainly not ready to abandon the use of a pre-intubation checklist based upon this study
Show References
Since Christmas is coming up, let's talk about Hemophilia A (factor VIII deficiency) and Hemophilia B (factor IX deficiency, also known as Christmas disease)
Deficiencies in Factors VIII and IX are the most common severe inherited bleeding disorders.
Pathophysiology:
- Factors VIII and IX are required for activation of factor X.
- In patients with Hemophilia A (factor VIII deficiency) or Hemophilia B (factor IX deficiency, also known as Christmas disease), after an injury, clot formation is delayed.
- Inadequate thrombin generation leads to failure to form a tightly crosslinked fibrin clot to support platelet plug, which leads to easy bleeding.
- Clot that is formed may be friable and rebleeding occurs during physiologic lysis of clots or with minimal new trauma
Clinical Manifestations:
- 2% of neonates with hemophilia have intracranial hemorrhages
- 30% of male infants with hemophilia bleed with circumcision
- Continued bleeding from umbilical stump in neonate
- In absence of positive family history (hemophilia has high rate of spontaneous mutation), hemophilia may go undiagnosed in a newborn
- Easy bruising, intramuscular hematomas, and hemarthroses (hallmark for hemophilic bleeding) begin when child begins to cruise
- Bleeding from minor traumatic lacerations of the mouth (e.g. torn frenulum) can persist for hours or days
- Iliopsoas hemorrhage: patient may lose large volumes of bleed into the muscle, leading to hypovolemic shock, with only a vague complaint of area of referred pain in the groin. Hip is held in a flexed, internally rotated position, due to irritation of the iliopsoas.
- Confirmed on CT or US
- Clinically unable to extend hip
- Hemarthrosis rare in patients with acquired hemophilia
Lab findings and diagnosis
- Reduced levels of factor VIII or factor IX will cause higher PTT
- PTT is usually 2-3x upper limit of normal in patients with severe hemophilia.
- Platelet count, bleeding time, prothrombin time, and thrombin time are all normal
- If PTT is not corrected after administration of factor VIII or IX, an inhibitor may be present.
- 25-35% of patients with hemophilia who received infusions of factor VIII or factor IX, a factor specific antibody may develop (inhibitor)
Genetics
- Hemophilia occurs in 1:5000 males, with 85% having factor VIII deficiency and 10-15% having factor IX deficiency
- No apparent racial predilection, appearing in all ethnic groups
Classification
- Severe hemophilia: <1% activity of specific clotting factor and bleeding is often spontaneous
- Moderate hemophilia: 1-5% activity and require mild
trauma to induce bleeding
- Mild hemophilia: >5% activity and can go many years before diagnosis and usually require significant trauma to induce bleeding.
Treatment
- Ask patient or family if they brought their dosing information with them or their factor replacement with them. In many cases, they have it!
- For life-threatening or major hemorrhages, dose should aim to achieve levels of 100% activity
- Hemophilia A: 50U/kg recombinant Factor VIII (each U/kg of factor VIII in hemophilia A increases factor by 2%)
- Hemophilia B: 100U/kg recombinant Factor IX (each U/kg of factor VIII in hemophilia A increases factor by 1%)
- Aim for 50% correction in moderate bleeds and 100% correction in severe bleeds
- If you don’t have factor-specific products:
- Hemophilia A
- can give 1U cryoprecipitate (~80U of factor VIII) or try PCC (as it contains factors II, VII, IX, and X)
- activated PCC (FEIBA) 75-100U/kg
- Hemophilia B
- FFP NO LONGER RECOMMENDED (volume of FFP required has high risk of volume overload)
- Cryoprecipitate does NOT contain factor IX, so will not work.
- Hemophilia A
- For acute bleeding in patients with mild hemophilia A:
- Can give DDAVP: increases factor VIII by 3-5x by encouraging release of endogenous factor VIII. Recommended dose: 0.3mcg/kg/dose IV
- For mild bleeding:
- TXA (clot stabilizer)
- Desmopressin
- Aminocaproic acid
- If patient has inhibitors:
- Hemophilia A:
- Activated PCC (75-100U/kg) (do NOT give if on patient is on emicizumab (Hemlibra) due to risk of thrombosis)
- Recombinant factor VII 90mcg/kg
- Hemophilia B:
- Recombinant factor VII 90mcg/kg
- Hemophilia A:
Summary:
- Aim for 50% correction in moderate bleeds and 100% correction in severe bleeds
- Hemophilia A: 50U/kg recombinant Factor VIII (each U/kg of factor VIII in hemophilia A increases factor by 2%)
- Hemophilia B: 100U/kg recombinant Factor IX (each U/kg of factor VIII in hemophilia A increases factor by 1%)
- Treatment if patient has no inhibitors:
- Hemophilia A:
- Severe bleed: Give full dose factor XIII (50U/kg), even if the patient is on prophylaxis
- Mild bleeds: factor XIII replacement (25U/kg), TXA, DDAVP, aminocaproic acid
- Hemophilia B:
- Severe bleed: Give full dose factor IX (100U/kg), even if the patient is on prophylaxis
- Mild bleeds: factor IV replacement (50U/kg), TXA, aminocaproic acid
- Hemophilia A:
- Treatment if patient has inhibitors:
- Hemophilia A:
- Activated PCC (75-100U/kg) (do NOT give if on patient is on emicizumab (Hemlibra) due to risk of thrombosis)
- Recombinant factor VII 90mcg/kg
- Hemophilia B:
- Recombinant factor VII 90mcg/kg
- Hemophilia A:
Show References
Since Christmas is coming up, let's talk about Hemophilia A (factor VIII deficiency) and Hemophilia B (factor IX deficiency, also known as Christmas disease)
Deficiencies in Factors VIII and IX are the most common severe inherited bleeding disorders.
Pathophysiology:
- Factors VIII and IX are required for activation of factor X.
- In patients with Hemophilia A (factor VIII deficiency) or Hemophilia B (factor IX deficiency, also known as Christmas disease), after an injury, clot formation is delayed.
- Inadequate thrombin generation leads to failure to form a tightly crosslinked fibrin clot to support platelet plug, which leads to easy bleeding.
- Clot that is formed may be friable and rebleeding occurs during physiologic lysis of clots or with minimal new trauma
Clinical Manifestations:
- 2% of neonates with hemophilia have intracranial hemorrhages
- 30% of male infants with hemophilia bleed with circumcision
- Continued bleeding from umbilical stump in neonate
- In absence of positive family history (hemophilia has high rate of spontaneous mutation), hemophilia may go undiagnosed in a newborn
- Easy bruising, intramuscular hematomas, and hemarthroses (hallmark for hemophilic bleeding) begin when child begins to cruise
- Bleeding from minor traumatic lacerations of the mouth (e.g. torn frenulum) can persist for hours or days
- Iliopsoas hemorrhage: patient may lose large volumes of bleed into the muscle, leading to hypovolemic shock, with only a vague complaint of area of referred pain in the groin. Hip is held in a flexed, internally rotated position, due to irritation of the iliopsoas.
- Confirmed on CT or US
- Clinically unable to extend hip
- Hemarthrosis rare in patients with acquired hemophilia
Lab findings and diagnosis
- Reduced levels of factor VIII or factor IX will cause higher PTT
- PTT is usually 2-3x upper limit of normal in patients with severe hemophilia.
- Platelet count, bleeding time, prothrombin time, and thrombin time are all normal
- If PTT is not corrected after administration of factor VIII or IX, an inhibitor may be present.
- 25-35% of patients with hemophilia who received infusions of factor VIII or factor IX, a factor specific antibody may develop (inhibitor)
Genetics
- Hemophilia occurs in 1:5000 males, with 85% having factor VIII deficiency and 10-15% having factor IX deficiency
- No apparent racial predilection, appearing in all ethnic groups
Classification
- Severe hemophilia: <1% activity of specific clotting factor and bleeding is often spontaneous
- Moderate hemophilia: 1-5% activity and require mild
trauma to induce bleeding
- Mild hemophilia: >5% activity and can go many years before diagnosis and usually require significant trauma to induce bleeding.
Treatment
- Ask patient or family if they brought their dosing information with them or their factor replacement with them. In many cases, they have it!
- For life-threatening or major hemorrhages, dose should aim to achieve levels of 100% activity
- Hemophilia A: 50U/kg recombinant Factor VIII (each U/kg of factor VIII in hemophilia A increases factor by 2%)
- Hemophilia B: 100U/kg recombinant Factor IX (each U/kg of factor VIII in hemophilia A increases factor by 1%)
- Aim for 50% correction in moderate bleeds and 100% correction in severe bleeds
- If you don’t have factor-specific products:
- Hemophilia A
- can give 1U cryoprecipitate (~80U of factor VIII) or try PCC (as it contains factors II, VII, IX, and X)
- activated PCC (FEIBA) 75-100U/kg
- Hemophilia B
- FFP NO LONGER RECOMMENDED (volume of FFP required has high risk of volume overload)
- Cryoprecipitate does NOT contain factor IX, so will not work.
- Hemophilia A
- For acute bleeding in patients with mild hemophilia A:
- Can give DDAVP: increases factor VIII by 3-5x by encouraging release of endogenous factor VIII. Recommended dose: 0.3mcg/kg/dose IV
- For mild bleeding:
- TXA (clot stabilizer)
- Desmopressin
- Aminocaproic acid
- If patient has inhibitors:
- Hemophilia A:
- Activated PCC (75-100U/kg) (do NOT give if on patient is on emicizumab (Hemlibra) due to risk of thrombosis)
- Recombinant factor VII 90mcg/kg
- Hemophilia B:
- Recombinant factor VII 90mcg/kg
- Hemophilia A:
Summary:
- Aim for 50% correction in moderate bleeds and 100% correction in severe bleeds
- Hemophilia A: 50U/kg recombinant Factor VIII (each U/kg of factor VIII in hemophilia A increases factor by 2%)
- Hemophilia B: 100U/kg recombinant Factor IX (each U/kg of factor VIII in hemophilia A increases factor by 1%)
- Treatment if patient has no inhibitors:
- Hemophilia A:
- Severe bleed: Give full dose factor XIII (50U/kg), even if the patient is on prophylaxis
- Mild bleeds: factor XIII replacement (25U/kg), TXA, DDAVP, aminocaproic acid
- Hemophilia B:
- Severe bleed: Give full dose factor IX (100U/kg), even if the patient is on prophylaxis
- Mild bleeds: factor IV replacement (50U/kg), TXA, aminocaproic acid
- Hemophilia A:
- Treatment if patient has inhibitors:
- Hemophilia A:
- Activated PCC (75-100U/kg) (do NOT give if on patient is on emicizumab (Hemlibra) due to risk of thrombosis)
- Recombinant factor VII 90mcg/kg
- Hemophilia B:
- Recombinant factor VII 90mcg/kg
- Hemophilia A:
Show References
Show References
Hot off the NEJM press, published November 11, 2023:
Restrictive or Liberal Transfusion Strategy in Myocardial Infarction and Anemia (Carson, Brooks, Hebert, et al Myocardial Ischemia and Transfusion (MINT) Investigators)
- Phase 3 interventional trial
- Adult patients with STEMI or NSTEMI and Hgb <10 g/dL were randomly assigned to either of two strategies
- restrictive transfusion- threshold of 7-8 g/dL (transfusion was permitted but not required when the hemoglobin level was less than 8 and was strongly recommended when the level was less than 7 or when anginal symptoms were not controlled with medications)
- liberal transfusion- threshold of 10 g/dL
- Primary outcome measured was recurrent MI or death at 30 days
3504 pateints were included in this study. A primary-outcome event occurred in 295 of 1749 patients (16.9%) in the restrictive-strategy group and in 255 of 1755 patients (14.5%) in the liberal-strategy group. More specifically, death occurred in 9.9% of the patients with the restrictive strategy and in 8.3% of the patients with the liberal strategy; myocardial infarction occurred in 8.5% and 7.2% of the patients, respectively.
Conclusions from the study: Liberal transfusion strategy did NOT significantly reduce the risk of recurrent MI or death at 30 days.
Show References
A chart review of 1300 patients over age 65 admitted to the trauma service, arrived as a trauma activation, or had an injury severity score over 12 over a 6 year period looking at 30 day mortality found:
"five factors associated with increased 30-day mortality in older trauma patients: GCS < 15, ISS > 15, age ≥ 85 years, anticoagulation, and multimorbidity."
Fall from standing was the leading cause of trauma
Again, fragility is the index we should be using, not age alone. This study is limited in its retrospective chart review nature. Prospective research in the area of geriatric trauma is needed. Until then, assess those over age 65 for risk factors associated with fragility and treat accordingly.
Show References
Show References
This retrospective study looked at trauma patients over age 65 and divided them into age ranges 65-80 (geriatric) and 80 plus (super-geriatric). They then looked at mechanusm of injury, mortality, interventions,etc. What they found was ages 65-80 were more likely to be injured in motor vehicle crashes vs. falls for those over 80. Those over 80 received less interventions including hemmorhage control surgery and had much higher levels of withdrawal of care.
This study highlights that the geriatric population is not as a monolithic group over age 65, but more nuanced by various age ranges over 65. Research going forward should be adjusted to these nuanced age ranges. Out treatment approaches should be adjusted in geriatric vs. super-geriatric patients as well.
Show References
A recently published study of ED APPs, residents, attendings, and nurses attempted to assess clinician's perspectives on how ED boarding impacts ED staff and patients. Authors performed a survey followed by focus group sessions to obtain qualitative insignts from participants.
All respondents associated boarding with feelings of burnout and self-reported poor satisfaction with communication and the process of boarding care.
Several key themes emerged which are outlined below:
- Clinicians perceived that boarding leads to increased patient safety events.
- Clinicians desired standardization for the boarding care process.
- Clinicians felt they had a lack of knowledge, resources, and training to care for boarding patients.
- Clinicians desired proactive bed and resource planning for boarding patients.
- Clinicians advocated for improved communication among the team and to patients.
- Clinicians identified a need for culture change regarding boarding care.
This publication highlights the negative workforce and patient safety effects of ED boarding. It amplifies the voices of our colleagues who work towards change to improve both the health of our wrokforce as well as that of our patients and the communities that we serve.
Show References
Acute bronchiolitis (AB) is a common cause of respiratory tract infections in infants. A recent study looked at the application of Point-of-Care Lung Ultrasound (LUS) in infants <12 months who presented with symptoms of AB.
They scored infant lungs using a cumulative 12-zone system. With the below scale:
0 - A lines with <3 B lines per lung segment.
1 - ≥3 B lines per lung segment, but not consolidated.
2 - consolidated B lines, but no subpleural consolidation.
3 - subpleural consolidation with any findings scoring 1 or 2.
They found that infants with higher LUS scores had increased rates of hospitalization and length of stay.
Here are some tips for ultrasounding a pediatric patient:
- Attempt to warm the gel
- Have the parent/relative hold the patient while scanning
- For those old enough, allow the child to investigate the ultrasound probe prior to placing the probe on the child.
- Destract the child while performing scanning
Show References
This small study suggests using reverse shock index times the Glasgow Comma Scake score may give a prognostication on pediatric trauma severity and resource utilization.
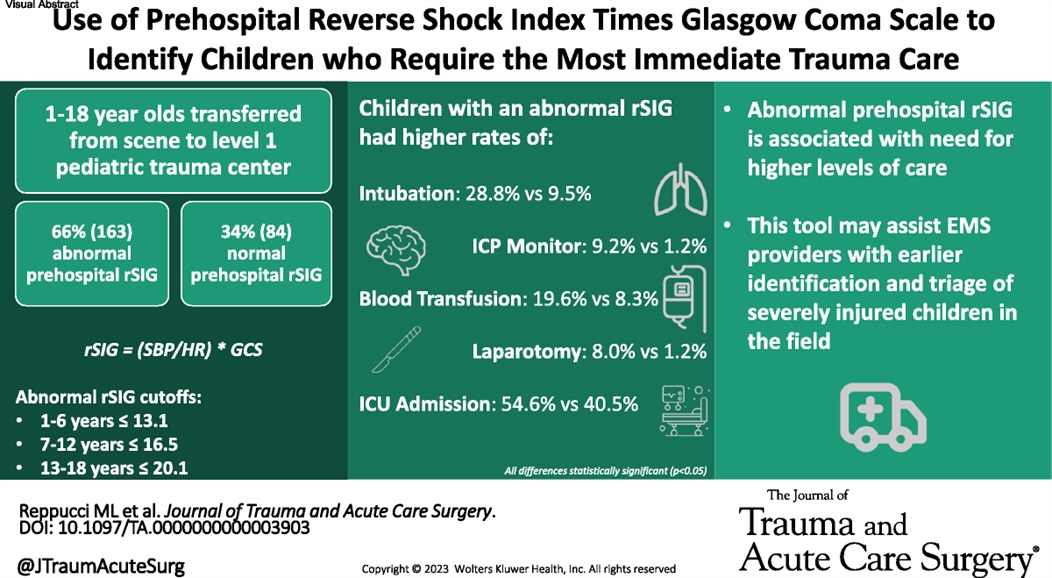
Show References
Show References
Show References
This pediatric ICU study measured ICP during and after ketamine infusion. There was no increase in ICP associated with the ketamine infusion. This small study adds to the data that ketamine is safe in pediatric brain injured patients.
Show References
Question
https://prod-images-static.radiopaedia.org/images/626179/d58f35a8aa4a0a6750a6adce4087a4_gallery.jpg
"I was kicked in the inside of my knee while it was straight (extended). Look at the x-ray and tell me if its bad"
Show Answer
For the folks who have been in practice for a while, you may be aware of the roller-coaster evidence base looking at steroids for pneumonia. Once thought to be beneficial and clearly indicated, of late steroids for pneumonia have fallen out of favor. Hamad et al have published an excellent (and brief) review in Clinical Infectious Diseases which suggests the pendulum might be swinging back in favor of giving steroids to patients with pneumonia. It's a ~5 minute read, so I recommend glancing through it yourself, but below are my two cents (solely my opinion) on where we are with steroids for pneumonia.
Take Home Points (OPINION ALERT):
1) When you have a condition present that you consider an indication for steroids (e.g. severe COVID-19 for sure; septic shock, s. pneumo infection, and ARDS depending on how you feel about the existing literature) --> strongly consider giving steroids unless there's a contraindication
2) When you have an undifferentiated patient who MAY have one of these conditions (e.g. pneumonia with COVID pending, patient potentially in ARDS or high risk of going into ARDS, etc) who is very sick --> it is reasonable to give steroids (if no contraindication) or not give steroids. My tendency is to lean towards giving steroids in these cases, but do be aware that society guidelines recommend against steroids here (although debatable if they just haven't caught up to more recent literature)
3) When you have an undifferentiated patient who may have one of these conditions, but is NOT very sick --> I do not think there is significant enough evidence to support empiric steroids
4) Factors that might push you one way or another:
- Severity of disease (more severe favors giving steroids),
- Pathogen (COVID-19 and s. pneumo favor steroids),
- What formulation of steroids you have availabile. Some of these studies used continuous hydrocortisone infusions, for example, which most hospitals don't routinely do.
- Comorbidities (uncontrolled diabetes, wound healing issues, risk for opportunistic infections might argue against giving steroids)
