Search
The RECOVERY (Randomized Evaluation of COVid-19 thERapY) investigators recently published a non-peer reviewed article on their findings utilizing dexamethasone to treat patients with COVID-19.
Rx: Dexamethasone 6mg daily* x 10 days (PO or IV) *or steroid equivalent
- 2104 in the dexamethasone group vs 4321 in the “usual care” group
- Did not exclude children or pregnant/breastfeeding mothers
- Follow-up at 28 days, hospital discharge, or death
Primary outcome: All-cause mortality at 28-days
Secondary outcomes:
- Major arrhythmia
- Time to discharge from hospital
- Duration of mechanical ventilation
- Need for renal replacement therapy
- In patients not ventilated at enrollment, need for intubation/ECMO & death
Results:
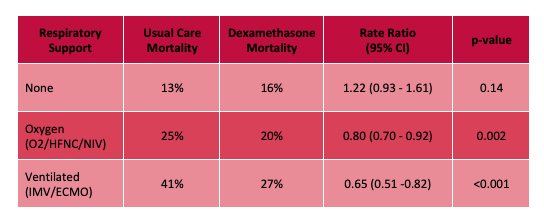
- Decrease in overall mortality at 28-days with 3% absolute risk reduction.
- NNT of 25 in patients requiring O2, HFNC, or NIV
- NNT of 8 in patients requiring invasive mechanical ventilation
- More mortality benefit seen the higher the respiratory support required, with no benefit and apparent trend towards increased mortality in the group not requiring any respiratory support at all.
- When stratified by symptoms < or > 7 days, mortality benefit only seen in the >7 days group (which was more of the ventilated patients).
- Less progression to intubation, shorter hospital duration, greater likelihood of hospital discharge.
Limitations:
- Not yet peer-reviewed, haven't seen all the data, additional analyses could be helpful in determining if treatment effect is real
- Unblinded study
- 7% of control group received dexamethasone
Bottom Line: Strongly consider admininstering dexamethasone to your patients with known COVID-19 who require respiratory support, and look for the peer-reviewed publication from the RECOVERY Trial investigators.
References
Horby P, Lim WS, Emberson et al. Effect of Dexamethasone in Hospitalized Patients with COVID-19: Preliminary Report. https://www.medrxiv.org/content/10.1101/2020.06.22.20137273v1 (Accessed July 14th, 2020)
