Neurological Injuries to the Brain after Cardiac Arrest
 Cardiac Arrest (CA) is a leading cause of death in the United States and the world. It causes 460,000 sudden cardiac deaths in the U.S. annually and 17 million worldwide.
Cardiac Arrest (CA) is a leading cause of death in the United States and the world. It causes 460,000 sudden cardiac deaths in the U.S. annually and 17 million worldwide.
Despite advances in resuscitation, survival after out‑of‑hospital cardiac arrest remains very low, with only about two to nine percent of patients surviving to hospital discharge.
Among survivors, poor neurological recovery, including prolonged coma or severe disability, is common and places a substantial burden on families and society. Neurological injury is also a leading cause of death in this population.
Our Research
We develop regenerative strategies to tip that balance toward recovery after cardiac arrest-induced global ischemic brain injury. We focus on metabolically glycoengineered neural stem cells (MGE‑NSCs) and optimized delivery routes after experimental cardiac arrest models to enhance brain repair.
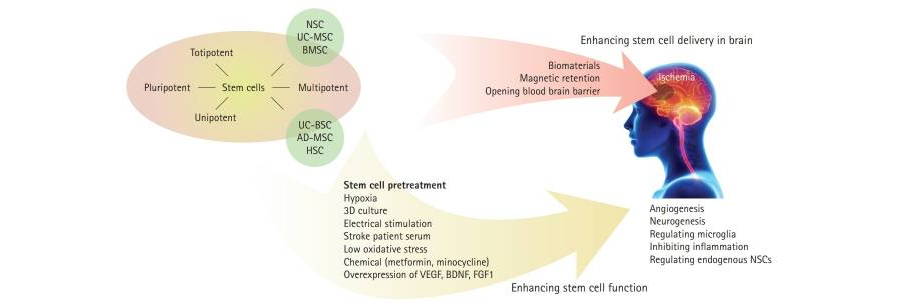
Using preclinical asphyxial cardiac arrest models, we also study how these treatments shape post-ischemic brain states and influence the success of cell-based therapies. Within this framework, we test how neural stem cell therapies and their extracellular vesicles, delivered at defined doses, through optimized routes, and at phase-specific time points after resuscitation to promote neuronal survival, circuit remodeling, and functional recovery.
Together, these studies aim to integrate advanced stem cell engineering and cell-based or cell-free approaches into phase-specific treatment strategies to enhance brain recovery after cardiac arrest.
Zhang, S., Lachance, B. B., Moiz, B., & Jia, X. (2020). Optimizing Stem Cell Therapy after Ischemic Brain Injury. Journal of Stroke, 22(3), 286-305.
Metabolic Glycoengineering (MGE) of Stem Cells to Enhance Brain Repair
Research Highlight
UMSOM News (May 2024)
Stem Cell Therapy Boosts Natural Repair After Cardiac Arrest
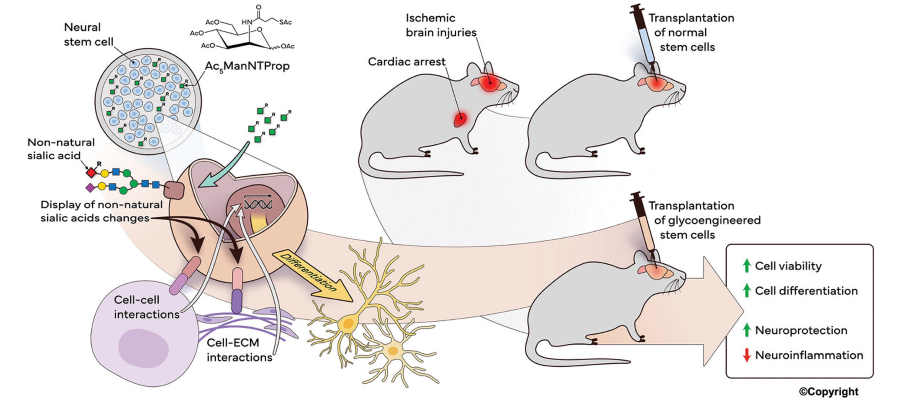
Metabolic glycoengineering (MGE) of neural stem cells is used to modulate cell-surface glycosylation in ways that enhance cell survival, integration, and reparative capacity following cardiac arrest-induced brain injury.
Pretreatment of human neural stem cells (hNSCs) with a synthetic ManNAc analog (Ac₅ManNTProp) remodels surface sialylation, leading to increased cell viability, enhanced neuronal differentiation, and improved synaptic plasticity.
In preclinical models of global cerebral ischemia, these metabolically engineered cells reduce neuroinflammation and are associated with improved functional recovery compared with unmodified naive neural stem cells.

J. Du, X. Liu, S. Marasini, Z. Wang, K. Dammen-Brower, K. J. Yarema, X. Jia, Metabolically Glycoengineered Neural Stem Cells Boost Neural Repair After Cardiac Arrest. Adv. Funct. Mater. (front cover) 2024, 34, 2309866.
Effect of MGE-NSC Delivery Route on Brain Recovery Post-Cardiac Arrest
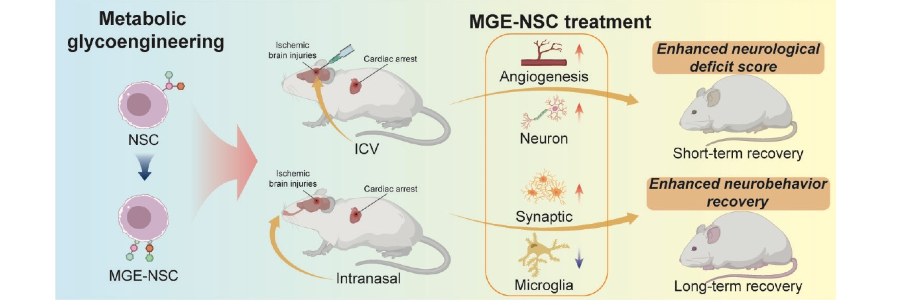
We compared intranasal and intracerebroventricular delivery of metabolically glycoengineered human neural stem cells as strategies for brain repair after cardiac arrest. Intranasal delivery, administered shortly after resuscitation, allowed engineered cells to access injured brain regions, promote vascular growth and neuronal differentiation, and support sustained recovery through a non-invasive route. Intracerebroventricular delivery produced stronger early neuroprotective effects, particularly in severe injury, but required a more invasive approach.
Ongoing studies continue to refine how delivery route, timing, and injury severity interact, with the goal of developing phase-specific stem cell strategies for post-cardiac arrest brain recovery.
Liu, X., Wang, Z., Du, J., Chae, S., Marasini, S., McElroy, M., Yarema, K. J., & Jia, X. (2025). Intranasal and intracerebroventricular delivery of metabolically glycoengineered neural stem cells to enhance post–cardiac arrest brain recovery. Neural Regeneration Research. (Advance online publication)
Intranasal Delivery of Stem Cell Therapy for Brain Injury After CA
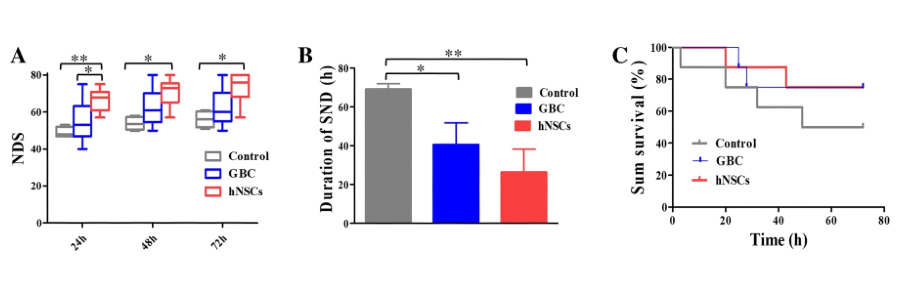
Figure: Rats that received either glibenclamide or human neural stem cells had better neurological scores after cardiac arrest than untreated rats, and the stem cell-treated group improved the most. The treatments also shortened the time animals remained in a severely impaired state, with stem cells again showing the greatest reduction.
We evaluate and compare the therapeutic effects of NSC and glibenclamide (GBC) therapies post-CA, and investigate mechanisms via Neuroinflammation Regulation that protect and preserve brain function after cardiac arrest. Rather than simply replacing lost neurons, our work demonstrates that transplanted stem cells actively modulate the post-ischemic brain microenvironment by limiting cell death, attenuating neuroinflammation, and activating endogenous repair pathways.
Neurological-deficit scores (NDSs) showed significant improvement after CA in hNSC and GBC groups compared to controls, and hNSC treatment was superior to GBC treatment. Through early intranasal administration, we define how stem cells enhance survival, targeting, and pro-repair signaling to support durable neurological recovery, with displayed superior inflammatory regulation in hNSC treatment.
Wang, Z., Zhang, S., Du, J. et al. Neuroprotection of NSC Therapy is Superior to Glibenclamide in Cardiac Arrest-Induced Brain Injury via Neuroinflammation Regulation. Transl. Stroke Res. 14, 723–739 (2023).
Intracerebroventricular Administration of hNSCs for Neurological Recovery
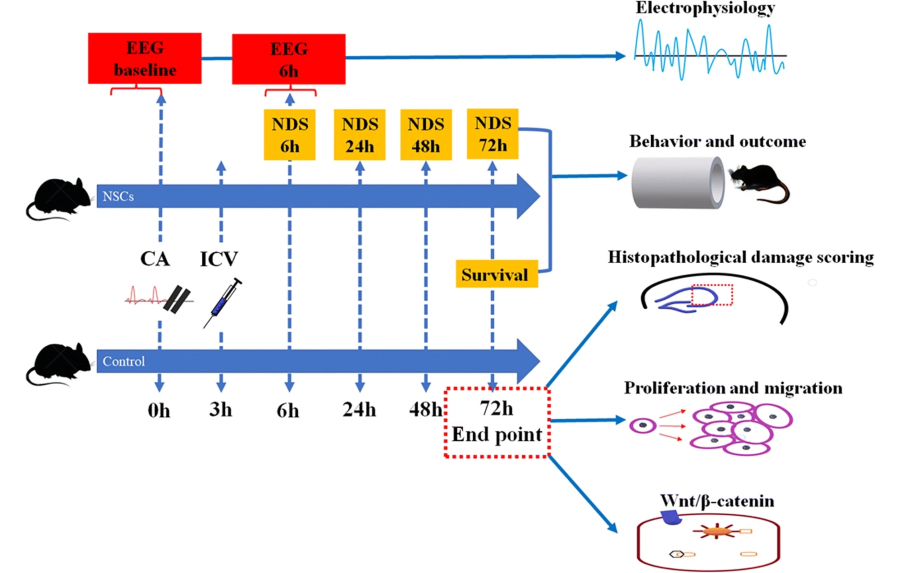
We developed stem cell-based strategies to repair the brain after cardiac arrest. In this study, we delivered human neural stem cells into the lateral ventricle of rats three hours after cardiac arrest and found that treated animals exhibited improved neurological deficit scores, reduced hippocampal neuronal damage, and earlier recovery of brain electrical activity compared with controls.
Mechanistically, intracerebroventricular hNSC transplantation enhanced proliferation and long-range migration of neural stem and progenitor cells tracked by Dcx antibody from the subventricular zone and activated Wnt/β-catenin signaling, supporting a synergistic model in which transplanted cells both replace lost neurons and stimulate intrinsic repair programs.
Wang, Z., Du, J., Lachance, B. B., Mascarenhas, C., He, J., & Jia, X. (2021). Intracerebroventricular Administration of hNSCs Improves Neurological Recovery after Cardiac Arrest in Rats. Stem cell reviews and reports, 17(3), 923–937.
Quantitative Monitoring of Cerebral Blood Flow for Targeted Temperature Management for Brain Recovery Post-Cardiac Arrest
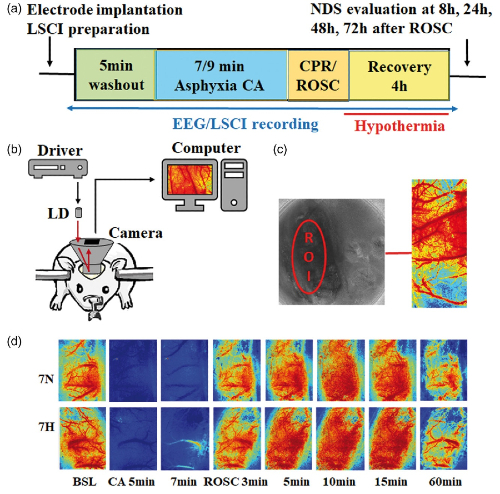 We develop and apply advanced optical imaging tools to understand how cerebral blood flow evolves after cardiac arrest and how targeted temperature management shapes brain recovery.
We develop and apply advanced optical imaging tools to understand how cerebral blood flow evolves after cardiac arrest and how targeted temperature management shapes brain recovery.
We used a custom laser speckle contrast imaging system to track relative blood flow at the level of individual arteries, veins, and capillaries in rats after asphyxial cardiac arrest under normothermic or hypothermic conditions.
Immediate hypothermic temperature management prolonged the early hyperemic phase, delayed and lowered subsequent hypoperfusion, and was associated with better EEG recovery and higher neurologic deficit scores, highlighting cerebral blood flow as a dynamic, mechanistic link between temperature therapy and neuroprotection after cardiac arrest.
He, J., Lu, H., Young, L., Deng, R., Callow, D., Tong, S., & Jia, X. (2019). Real-time quantitative monitoring of cerebral blood flow by laser speckle contrast imaging after cardiac arrest with targeted temperature management. Journal of cerebral blood flow and metabolism: official journal of the International Society of Cerebral Blood Flow and Metabolism, 39(6), 1161–1171.
Figure: Diagram of laser speckle contrast imaging (LSCI) used to track brain blood flow in rats after cardiac arrest. The setup records color maps of cortical blood flow over time, showing how perfusion changes from baseline through arrest and recovery under normothermia (7N) and hypothermia (7H).
