Delivering Targeted Biological Support Directly to Injured Tissue
 Jiang, L., Mee, T., Zhou, X., & Jia, X. (2021). Augmenting peripheral nerve regeneration with adipose-derived stem cells. Stem Cell Reviews and Reports, 18(2), 544–558.
Jiang, L., Mee, T., Zhou, X., & Jia, X. (2021). Augmenting peripheral nerve regeneration with adipose-derived stem cells. Stem Cell Reviews and Reports, 18(2), 544–558.
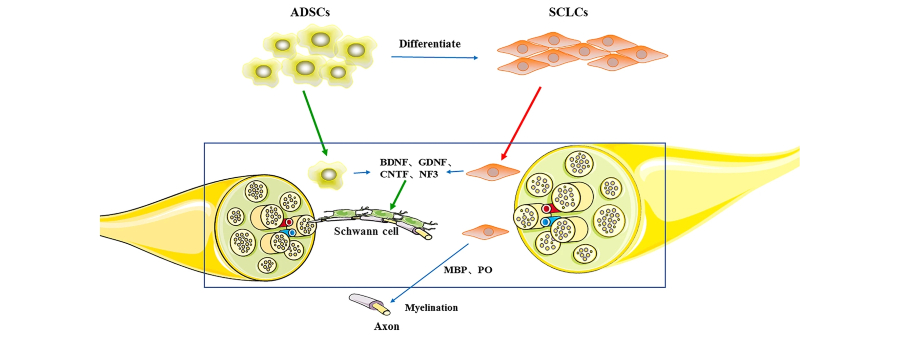
Peripheral nerve injuries affect millions of people worldwide and often result in lasting weakness, numbness, or chronic pain when natural repair is incomplete. Our lab’s primary focus is on developing new therapies, including stem cell–based therapies, to enhance nerve regeneration by delivering targeted biological support directly to injured tissue.
Using preclinical models of nerve injury, we study how therapeutic targets interact with Schwann cells and regenerating axons to promote growth, remyelination, and functional recovery. We developed these cell-based approaches, together with bioengineered nerve scaffolds and controlled growth factor delivery, to precisely regulate the spatial and temporal cues that guide repair.
In parallel, we explore complementary cell-free strategies, including extracellular vesicles, to harness regenerative signaling while advancing clinically translatable solutions for nerve repair after severe nerve injury.
Extracellular Vesicles for Peripheral Nerve Regeneration
 Dutta, D., Pirolli, N. H., Levy, D., Tsao, J., Seecharan, N., Wang, Z., Xu, X., Jia, X., & Jay, S. M. (2023). Differentiation state and culture conditions impact neural stem/progenitor cell-derived extracellular vesicle bioactivity. Biomaterials Science, 11, 5474–5489.
Dutta, D., Pirolli, N. H., Levy, D., Tsao, J., Seecharan, N., Wang, Z., Xu, X., Jia, X., & Jay, S. M. (2023). Differentiation state and culture conditions impact neural stem/progenitor cell-derived extracellular vesicle bioactivity. Biomaterials Science, 11, 5474–5489.
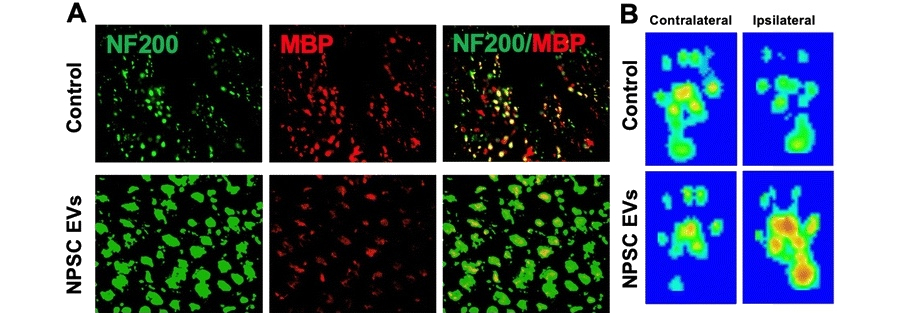
Figure 1: NPSC EVs enhance axonal regeneration and functional recovery after sciatic nerve crush injury. Representative images show increased NF200 and MBP co-labeled myelinated axons in NPSC EV–treated nerves compared to controls (A) and improved sciatic functional index heatmaps on the ipsilateral side following EV treatment (B).
We are developing cell-free, EV-based strategies to improve peripheral nerve repair, reduce muscle atrophy, and accelerate functional recovery after injury. Neural progenitor/stem cells (NPSCs)-derived extracellular vesicles were engineered under optimized culture conditions by tuning extracellular matrix cues and growth factor signaling, to enhance the therapeutic activity acting as a cell-free “mini-therapy” for improved nerve repair.
After fibronectin plus NGF was identified as the condition that maximized neurite outgrowth and anti-inflammatory activity in vitro, these EVs were administrated in a rat sciatic nerve crush model, leading to increased axon number and diameter, enhanced remyelination, improved sciatic functional index scores, and markedly preserved gastrocnemius muscle mass compared with controls, thereby demonstrating that significantly improved structural and functional recovery after peripheral nerve injury by carefully tuned neural stem cell EVs.
Optimized Electrical Stimulation Boost Stem Cell Therapy for Peripheral Nerve Regeneration
 Du J, Zhen G, Chen H, Zhang S, Qing L, Yang X, Lee G, Mao HQ, Jia X*. Optimal Electrical Stimulation Boosts Stem Cell Therapy in Nerve Regeneration. Biomaterials. 2018 Oct;181:347-359. doi: 10.1016/j.biomaterials.2018.07.015. Epub 2018 Jul 20. PMID: 30098570
Du J, Zhen G, Chen H, Zhang S, Qing L, Yang X, Lee G, Mao HQ, Jia X*. Optimal Electrical Stimulation Boosts Stem Cell Therapy in Nerve Regeneration. Biomaterials. 2018 Oct;181:347-359. doi: 10.1016/j.biomaterials.2018.07.015. Epub 2018 Jul 20. PMID: 30098570
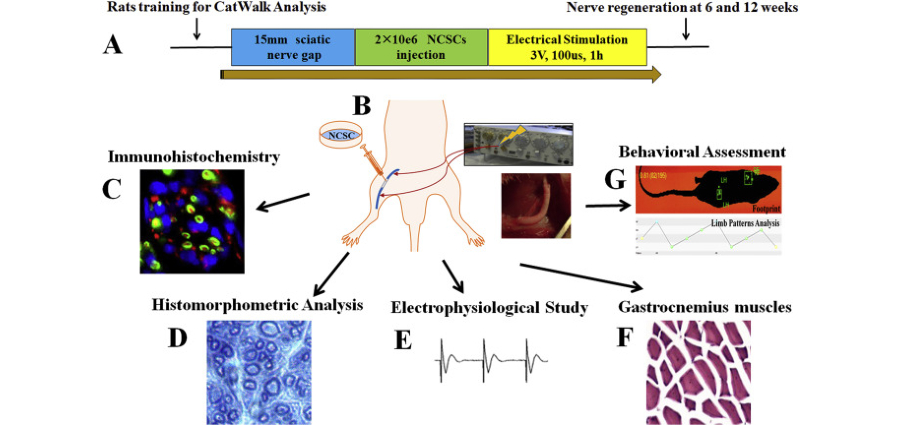
Human neural crest stem cell therapy was enhanced by precisely tuned brief electrical stimulation to repair critical 15 mm sciatic nerve gaps in rats. Optimized stimulation was shown to increase NCSC survival and to promote robust Schwann-like differentiation, resulting in denser myelinated axons with thicker myelin, larger axon diameters, and greater myelinated area than were observed in control conduits or NCSCs alone.
In vivo, nerve conduction signals were improved, gastrocnemius muscle mass and fiber morphology were restored, and improved gait metrics were produced by the combined NCSC + ES treatment, approaching or matching autograft repair while being outperformed by neither electrical stimulation nor NCSC transplantation alone.
Macrophage Activation after Peripheral Nerve Injury
 Xu, X.; Zhou, X.; Du, J.; Liu, X.; Qing, L.; Johnson, B.N.; Jia, X. Macrophage Activation in the Dorsal Root Ganglion in Rats Developing Autotomy after Peripheral Nerve Injury. Int. J. Mol. Sci. (2021). 22, 12801.
Xu, X.; Zhou, X.; Du, J.; Liu, X.; Qing, L.; Johnson, B.N.; Jia, X. Macrophage Activation in the Dorsal Root Ganglion in Rats Developing Autotomy after Peripheral Nerve Injury. Int. J. Mol. Sci. (2021). 22, 12801.
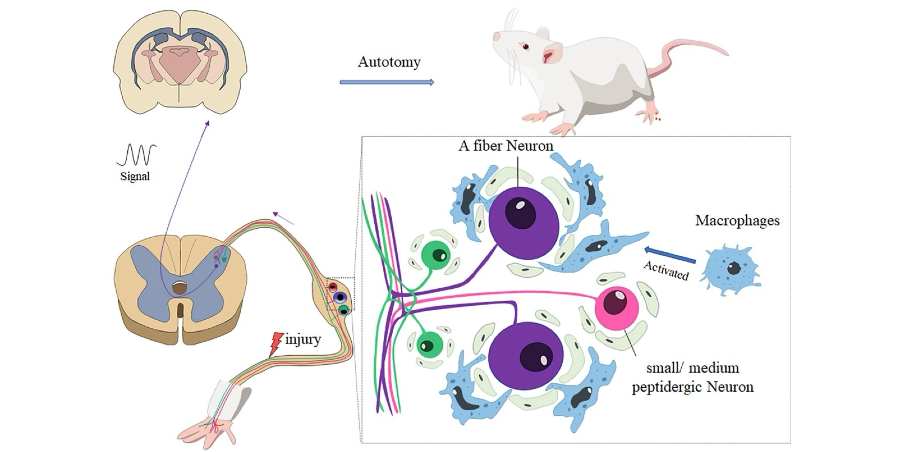
Autotomy of a denervated limb is common after peripheral nerve injury and was found to be markedly less frequent and less severe in nude rats than in Wistar rats over 12 weeks following sciatic nerve transection and repair. At both six and 12 weeks, significantly higher autotomy incidence and Walls scores were observed in Wistar rats, paralleling a larger and more persistent accumulation of activated Iba1⁺ macrophages in the dorsal root ganglion (DRG), an increased proportion of NF200⁺ myelinated A-fiber neurons, and dense clustering of activated macrophages around these A-fiber neurons, whereas CGRP⁺ peptidergic neurons were unchanged and CD4⁺ T cells were absent in both strains, suggesting autotomy is centrally mediated by macrophage–A-fiber interactions within the DRG.
Therapeutic Effects of Peripherally Administered Neural Crest Stem Cells
Neural crest stem cells (NCSCs)-loaded and cell-free scaffolds were evaluated for 12 weeks in complex sciatic nerve gap-injured rats. Catwalk gait analysis showed a significantly higher stand index and maximum intensity in the NCSC group. The mechanical and thermal allodynia in the ipsilateral hind paw were significantly relieved in the NSCS group, indicating a reduced development of neuropathic pain.
The astrocyte and microglial activation were decreased in the spinal cord, while activation of extracellular signal-regulated kinase (ERK) and NF-ᴋB P65 signals, and transient receptor potential vanilloid 1 (TRPV1) expression were downregulated. Peripherally-administered NCSC mitigated PNI-induced neuropathic pain, improved functional recovery, and protected the spinal cord from glial activation and central sensitization.
Personalized 3D‑Printed Nerve Guides for Complex Peripheral Nerve Regeneration
A 3D-printed, anatomically matched nerve guide was created to reconstruct complex, bifurcating peripheral nerve pathways such as the severe sciatic nerve. Using structured-light 3D scanning and microextrusion printing, conduits were produced in which native branch geometry was replicated, axon-aligning luminal microgrooves were presented, and path-specific growth factor gradients (NGF in the sensory channel and GDNF in the motor channel) were delivered to steer regenerating axons.
In a rat model with a 10 mm bifurcated sciatic nerve gap, robust axonal regrowth and Schwann cell–associated myelination in both branches were supported by gradient-functionalized guides, and significantly improved gait duty-cycle recovery was achieved compared with identical guides without gradients, thereby demonstrating that personalized, functionally effective peripheral nerve repair can be enabled through imaging-guided 3D printing.
Biodegradable Regenerative Neural Interfaces for Peripheral Nerves
Fully biodegradable neural interfaces are being created to guide peripheral nerve regeneration and to enable long-term recording of neural activity. Tunable, tyrosine-derived polymer materials were designed with mechanical properties and degradation rates aligned with sciatic nerve regrowth, and porous, multiluminal nerve conduits with integrated microelectrodes were fabricated from these materials.
In rabbit sciatic defect models, robust axonal ingrowth was supported by these regenerative interfaces, and stable, multi-channel neural recordings were provided, emerging within weeks and strengthening over time. This work lays the foundation for next-generation bioresorbable neural interfaces that could restore communication with the nervous system after limb loss. Active peripheral nerve repair can be enabled through imaging-guided 3D printing.
