Research
Our research can be divided into 2 broad areas. The first area involves chemical biology of vertebrate development, which entails discovery of small molecules that selectively modulate signaling pathways involved in embryogenesis. Because developmental pathways represent important untapped therapeutic targets, we have an active medicinal chemistry program to develop our novel small molecules as lead compounds for future therapies. We have thus far discovered potent and highly selective chemical modifiers of bone morphogenetic protein (BMP), Wnt, Hedgehog, and lipid signaling pathways, among others. Several of our compounds are first-in-class molecules with substantial therapeutic potential in rare and common diseases, including cancers, atherosclerosis and heart failure. Thus, our chemical biological exploration is leading to new opportunities for innovative therapeutic programs.
In the second, we are exploring the potential of patient-derived induced pluripotent stem cells (iPSC) to study and treat human heart diseases. We use human iPSCs as renewable cell sources for examining the fundamental cell biology and physiology of normal and diseased human cardiomyocytes. Finally, we collaborate with bioengineers to develop human iPSC-derived heart tissues as a platform for drug discovery and testing, including validation of novel cardiac inotropes.
Chemical Genetics
Chemical Genetics of Embryonic Development

In a manner analogous to classic mutagenesis screens, we conduct high-throughput chemical screens using zebrafish to discover small molecules that specifically perturb embryonic pattern formation. Using the interdisciplinary chemical genetic approach, we have discovered exquisitely selective modulators of the Bone Morphogenetic Protein (BMP), Wnt and Hedgehog pathways, as well as important new signaling components that direct early vertebrate development.
In addition, we have launched the Chemical Phenomics Initiative, through which we are making our chemical genetic tools available to the public with the goal of fostering collaborations, and accelerating discovery and translation.
For more information, visit Chemical Phenomics Initiative.
Therapeutic Discovery
 Therapeutic Discovery
Therapeutic Discovery
Because aberrant activities of developmental pathways play major roles in pathogenesis of many human diseases, we are developing our novel small molecules as probes to discover future therapies.
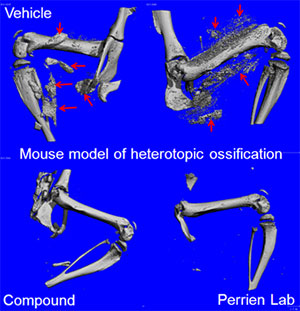
For example, we have made key contributions to the elucidation of BMP signaling as a promising therapeutic target for anemia of:
- chronic disease
- heterotopic ossification syndromes
- inflammatory bowel disease
- atherosclerosis
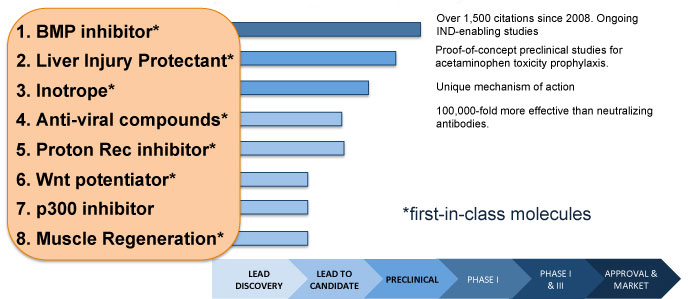
D2D Program Pipeline
Regenerative Chemical Biology
Regenerative Chemical Biology
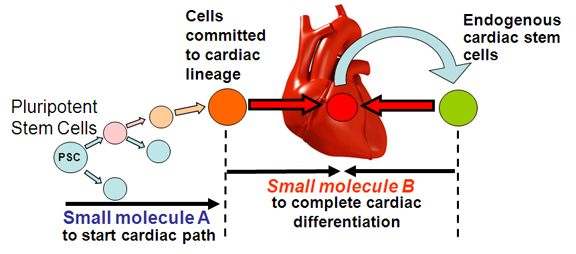
Since small molecules that perturb embryonic patterning do so by promoting acquisition of specific cell fates, we predict that they will be valuable chemical reagents for stem cell research and cell-based regenerative therapies.
For example, our small molecules are widely used for robust induction of neurogenesis and cardiomyogenesis in pluripotent stem cells.
Human Cardiomyocyte Biology
Human Cardiomyocyte Biology
We are exploring the potential of patient-derived induced pluripotent stem cells (iPSC)to study and treat human heart diseases. We use human iPSCs as renewable cell sources for examining the fundamental cell biology and physiology of normal and diseased human cardiomyocytes.
Finally, we collaborate with bioengineers to develop human iPSC-derived heart tissue.
Traditional hiPSC-CM
Mattress hiPSC-CM
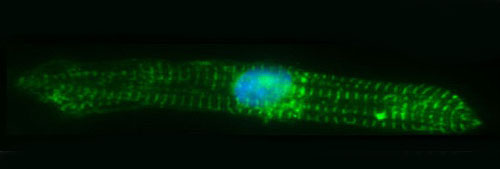
The Hong lab has also developed "Heart-on-a Dish" model for simultaneous calcium dynamic and myocyte contractility measurements of human iPSC-derived cardiomyocytes.
More on Human Cardiomyocyte Biology
Data Science
We have partnered with colleagues in the University of Maryland Baltimore County (UMBC) - Department of Mathematics and Statistics, NHLBI and Amish Research Program and Program for Personalized Genomic Medicine to utilize advanced Artificial Intelligence/Machine Learning as well as traditional methodologies to mine the wealth of data within electronic health records, UK Biobank and chemical phenomics initiative to reveal novel biological insights and to discover and validate therapeutic targets.
