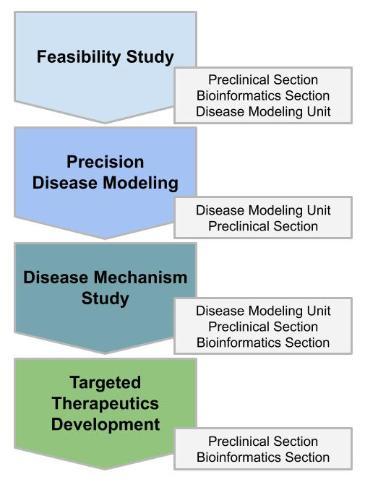
Four Levels of Service
The biomedical community already has identified tens of millions of genetic variants in the human genome, only a small percentage of which are expected to be causally associated with the disease. This creates an urgent need for high-throughput cost-effective means of obtaining functional data for these variants, especially those with clinical relevance. CPDM-UMSOM has developed a unique combination of Precision Disease modeling platforms that include a fly (Drosophila melanogaster), zebrafish (Danio rerio) and human-induced pluripotent stem cells (iPSC), to provide variant functional studies to clinicians and human geneticists working on a broad range of diseases. The biomedical community can nominate human genetic disease variants through this website, which also holds a catalog of our carefully curated and already available Precision Disease models as well as a host of additional information and resources. To best serve the community we have developed a 4-Level service pipeline that covers the entire process from community-nominated genetic variants to targeted therapeutics that benefit patients:
- Evaluate the feasibility of community-nominated variants
- Generate Precision Disease models to establish variant causal associations
- Discover the underlying disease mechanism in the Precision Disease model
- Develop and test targeted therapeutics using the Precision Disease models.
Level 1 Service: Feasibility Study
Given the millions of novel genetic variants already identified (and millions that are yet to come), it is critically important that we have an effective evaluation pipeline to go through large numbers of variants and identify the ones that are feasible for Precision Disease modeling. This is also extremely beneficial for the users since no one wants to pay for service that is likely to yield an uninformative result. The Preclinical and Bioinformatics Sections will play an essential role in this, to ensure that only the most highly feasible variants will be passed on to the Disease Modeling Unit in order to prevent the DMU from becoming overwhelmed due to the labor-intensive nature of their part in the service pipeline. After the CPDM receives a variant and associated clinical information, the Preclinical Section will first evaluate whether the quality of the clinical data is sufficient. If so, the variant will be passed to the Bioinformatics Section, where a sophisticated and comprehensive “filtering system” has been developed. This approach will combine a series of operations including searching large patient databases, performing linkage analysis, analyzing structural/functional relationships of specific variants, as well as, searching existing biomedical research literature for relevant genes, diseases, and variants. The result of our Level 1 service is a “Precision Modeling Feasibility Report” that will be offered to users of the biomedical community for free.
Level 2 Service: Precision Disease Modeling
For variants deemed feasible for Precision Disease modeling, the DMU will develop a proposal with a detailed description of how the Precision Disease model will be generated, and what phenotypic assays will be used to validate the causal association of the variants according to the patient medical problems. DMU will work with the Preclinical Section on this proposal to ensure that the phenotypic assays proposed are relevant to the clinical features of the patient. This proposal will be presented to the user by the Service Section as the “Precision Disease Modeling Proposal”. The DMU is also responsible to estimate the cost for the proposed work and generate a quote for this Level 2 service proposal.
If the user approves the “Precision Disease Modeling Proposal”, DMU will generate the proposed Precision Disease model and perform the clinically relevant phenotypic assays. DMU will consult the Preclinical Section to cross validate their results with patient medical records and to generate a Level 2 service report, our “Precision Disease Modeling Phenotypic Report”, as the product of this service. This report aims to provide in vivo functional evidence to validate (or revoke) the disease association of the variant under investigation. We will continue to improve the Precision Disease model generation and phenotypic assay development, including by developing new phenotypic assays based on user input. The Precision Disease models generated and validated will be passed to the Service Section for curating and cataloging to make them available for distribution to the user and the wider biomedical community via website orders fulfilled by the Service Section.
Level 3 Service: Disease Mechanism Study
If the results from the “Precision Disease Modeling Phenotypic Report” are promising, it might be feasible to continue study of the disease mechanism in order to ultimately develop targeted therapy. In this case, the DMU will work with the Preclinical Section (which is responsible for collecting patient omics and/or biomarker data), to generate the “Disease Mechanism Study Proposal” with a testable hypothesis (Level 3 service proposal).
Upon approval from the user, DMU will develop biomarkers that are geared to provide new insights into the underlying disease mechanism. The DMU will also work with the Preclinical Section and the user to develop new functional assays for the Precision Disease models that best reflect clinical testing in the patient; if applicable additional patient data might be obtained in collaboration with the physician. DMU will also generate relevant omics data from the Precision Disease models and pass these to the Bioinformatics Section to perform a cross-species comparison. Results will be shared and discussed with the Preclinical Section, summarized as the “Precision Disease Modeling Mechanism Study Report” and passed to the Service Section to share with the user as the product of the Level 3 service.
Level 4 Service: Targeted Therapeutics Development
If the results from the “Precision Disease Modeling Mechanism Study Report” suggest a potential therapeutic approach, the DMU will design a therapeutic screen using the CPDM Precision Disease models, and work closely with the Preclinical Core (which is tasked with toxicity studies as well as identification and selection of the best drug candidates) to generate the “Precision Disease Modeling Targeted Drug Study Proposal” (Level 4 Service proposal).
Upon user approval, DMU and Preclinical Section will perform and interpret the results of the proposed therapeutics screen. If needed, they will generate omics data to evaluate the treatment response and work with the Bioinformatics team to analyze the pharmacogenomics data. All data and results will be summarized as the “Precision Disease Modeling Targeted Drug Study Report” and presented to the user, by the Service Section, as the product of our Level 4 service.

